アウラノフィン 化学特性,用途語,生産方法
外観
白色~ほとんど白色、結晶性粉末
溶解性
アセトンに溶けやすく、エタノールにやや溶けにくく、水にほとんど溶けない。
用途
自己抗体産生及び炎症性細胞 機能抑制作用を示します。
用途
自己抗体産生及び炎症性細胞
機能抑制作用を示します。
効能
抗リウマチ薬
説明
Auranofin is the first orally effective gold compound to be marketed for the
treatment of severe rheumatoid arthritis. It is better tolerated and more
convenient than gold sodium thiomalate, which is administered intramuscularly.
化学的特性
White to Off-White Solid
使用
Auranofin is a new oral gold-based antiarthritis drug. Auranofin inhibits various leukocyte activation pathways at multiple sites. Auranofin inhibits the release of inflammatory mediators from human m
acrophages, basophils, and pulmonary mast cells. Auranofin is an efficient inducer of mitochondrial membrane permeability transition pore in the presence of calcium ions related to its inhibition of m
itochondrial thioredoxin reductase.
定義
ChEBI: An S-glycosyl compound consisting of 2,3,4,6-tetra-O-acetyl-1-thio-beta-D-glucopyranose with the sufur atom coordinated to (triethylphosphoranylidene)gold. It is administered orally fo
the treatment of active progressive rheumatoid arthritis.
応用例(製薬)
Auranofin ([tetra-O-acetyl-β-D- (glucopyranosyl)thio]-triethylphosphine)gold(I) is a second-generation gold-based drug, licensed as an orally available gold drug for the treatment of RA. It features a linear S Au P geometry, as shown by X-ray analysis. It is more lipophilic than the first-generation drugs, which makes oral administration possible. Treatment with Auranofin requires less visits to the clinic, but it is believed to be less successful in the treatment of RA compared to gold drugs being administered intramuscularly.
臨床応用
Auranofin is indicated in adults with active rheumatoid arthritis who have not responded sufficiently to one or more
NSAIDs.
安全性プロファイル
Poison by ingestion,intraperitoneal, and intravenous routes. Human systemiceffects by ingestion: ulceration or bleeding from stomach.An experimental teratogen. Other experimentalreproductive effects. Human mutation data reported.When heated to decomp
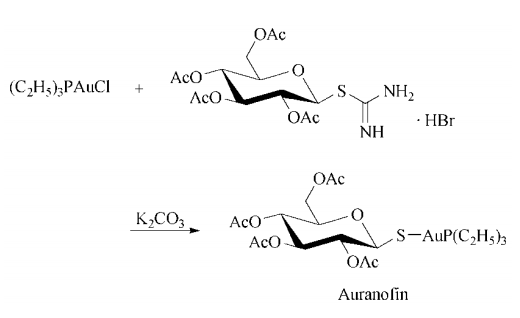
合成
Synthesis: ethanolic thiodiglycol is treated
first with aqueous gold(I) acid chloride trihydrate, then with ethanolic triethylphosphine to
give triethylphosphine gold(I) chloride, which is
added to an aqueous solution of S-(2,3,4,6-tetra-O-acetylglucopyranosyl)pseudothiourea hydrobromide and potassium carbonate to give the desired auranofin.
代謝
On a mg gold/kg basis, it is reported to be as effective in the rat adjuvant arthritis assay as the parenterally
effective drugs. Daily oral doses produce a rapid increase in kidney and blood gold levels for the first 3 days of
treatment, with a more gradual increase on subsequent administration. Plasma gold levels are lower than those
attained with parenteral gold compounds. The major route of excretion is via the urine. Auranofin may produce fewer
adverse reactions than parenteral gold compounds, but its therapeutic efficacy also may be less.
アウラノフィン 上流と下流の製品情報
原材料
準備製品