オピカポン
|
|
|
- CAS番号.
- 923287-50-7
- 化学名:
- オピカポン
- 别名:
- オピカポン;オピカポン (JAN);2,5-ジクロロ-3-[5-(3,4-ジヒドロキシ-5-ニトロフェニル)-1,2,4-オキサジアゾール-3-イル]-4,6-ジメチルピリジン1-オキシド;2,5-ジクロロ-3-[5-(3,4-ジヒドロキシ-5-ニトロフェニル)-1,2,4-オキサジアゾール-3-イル]-4,6-ジメチルピリジン-1-イウム-1-オラート;オンジェンティス錠;2,5-ジクロロ-3-[5-(3,4-ジヒドロキシ-5-ニトロフェニル)-1,2,4-オキサジアゾール-3-イル]-4,6-ジメチルピリジン=1-オキシド
- 英語名:
- Opicapone
- 英語别名:
- CS-1268;Opicapone;BIA 9-1067;Opicapone-d6;100G,500G,1KG;Opicapone (BIA 9-1067);Inhibitor,inhibit,COMT,Opicapone;5-[3-(2,5-Dichloro-4,6-dimethyl-1-oxido-3-pyridinyl)-1,2,4-oxadiazol-5-yl]-3-nitro-1,2-benzenediol;2,5-dichloro-3-(5-(3,4-dihydroxy-5-nitrophenyl)-1,2,4-oxadiazol-3-yl)-4,6-dimethylpyridine 1-oxide;1,2-Benzenediol, 5-[3-(2,5-dichloro-4,6-dimethyl-1-oxido-3-pyridinyl)-1,2,4-oxadiazol-5-yl]-3-nitro-
- CBNumber:
- CB82628219
- 化学式:
- C15H10Cl2N4O6
- 分子量:
- 413.17
- MOL File:
- 923287-50-7.mol
|
オピカポン 物理性質
- 沸点 :
- 701.1±70.0 °C(Predicted)
- 比重(密度) :
- 1.80±0.1 g/cm3(Predicted)
- 溶解性:
- DMSO : 100 mg/mL (242.03 mM; Need ultrasonic)H2O : < 0.1 mg/mL (insoluble)
- 外見 :
- Powder
- 酸解離定数(Pka):
- 4.67±0.38(Predicted)
安全性情報
- リスクと安全性に関する声明
- 危険有害性情報のコード(GHS)
| 絵表示(GHS) |

|
| 注意喚起語 |
警告 |
| 危険有害性情報 |
| コード |
危険有害性情報 |
危険有害性クラス |
区分 |
注意喚起語 |
シンボル |
P コード |
| H302 |
飲み込むと有害 |
急性毒性、経口 |
4 |
警告 |
 |
P264, P270, P301+P312, P330, P501 |
| H315 |
皮膚刺激 |
皮膚腐食性/刺激性 |
2 |
警告 |
 |
P264, P280, P302+P352, P321,P332+P313, P362 |
| H319 |
強い眼刺激 |
眼に対する重篤な損傷性/眼刺激 性 |
2A |
警告 |
 |
P264, P280, P305+P351+P338,P337+P313P |
| H335 |
呼吸器への刺激のおそれ |
特定標的臓器毒性、単回暴露; 気道刺激性 |
3 |
警告 |
 |
|
|
| 注意書き |
| P261 |
粉じん/煙/ガス/ミスト/蒸気/スプレーの吸入を避ける こと。 |
| P305+P351+P338 |
眼に入った場合:水で数分間注意深く洗うこと。次にコ ンタクトレンズを着用していて容易に外せる場合は外す こと。その後も洗浄を続けること。 |
|
オピカポン 価格
| メーカー |
製品番号 |
製品説明 |
CAS番号 |
包装 |
価格 |
更新時間 |
購入 |
オピカポン 化学特性,用途語,生産方法
効能
パーキンソン病治療薬, COMT阻害薬
説明
Opicapone is a selective and
reversible catechol O-methyltransferase (COMT) inhibitor that
was developed by the Portuguese pharmaceutical firm Bial and
sold to Neurocrine Biosciences. The drug was approved by
the USFDA as adjunctive treatment to levodopa (L-Dopa)/
dopa-decarboxylase inhibitor (DDCI) therapy in adults with
Parkinson’s disease (PD) and end-of-dose motor fluctuations
that cannot be stabilized on those combinations. In 14- to 15-
week double-blind multinational trials and in one-year openlabel
extension studies in this patient population, opicapone
was an effective and generally well-tolerated adjunctive therapy
to L-Dopa plus a DDCI and other PD therapies. During the
double-blind phase, adjunctive opicapone (50 mg once daily)
provided significantly greater improvements in motor fluctuations
than placebo, and no new unexpected safety concerns
were identified after treatment with opicapone over a 1.4 year
period. Furthermore, no serious cases of hepatotoxicity were
reported in clinical trials, which represents a significant safety
profile improvement over existing standard-of-care COMT
inhibitors enticapone, tolcapone, and nebicapone.
使用
Opicapone, is used for the synthesis of novel nitrocatechol-substituted heterocycles, having the ability to inhibit catechol-O-methyltransferase (COMT), used for the treatment of Parkinson`s diseases.
合成
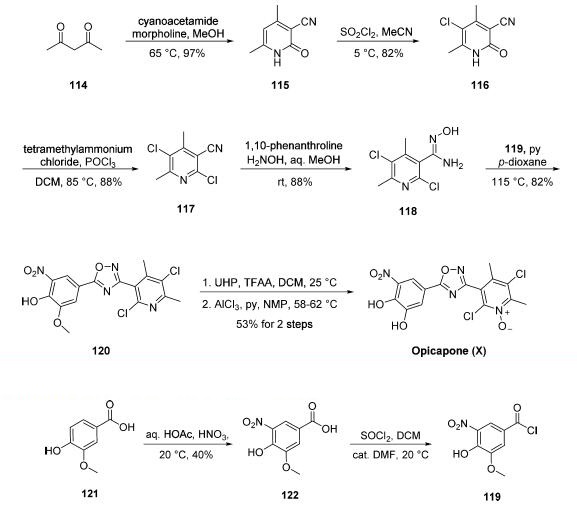
Although several synthetic approaches to opicapone or
opicapone subunits have been disclosed, a synthetic approach
described by Bial was exemplified on a scale capable of
producing 14.4 kg of the active pharmaceutical ingredient
(API). Commercial 2,4-pentanedione (114) was condensed
with cyanoacetamide in warm methanol to give rise to
cyanopyridone 115 in excellent yield. Chlorination
with sulfuryl chloride in chilled acetonitrile followed by
treatment with phosphorus oxychloride resulted in dichloropyridine
117. Next, treatment with hydroxylamine in aqueous
methanol converted nitrile 117 to the corresponding Nhydroxyamidine
118, and this was followed by exposure to
pyridine and acid chloride 119. These operations facilitated a cyclization
reaction, which furnished the key oxadiazole 120 in good yield.
Subjection of 120 to urea hydrogen peroxide (UHP) in
dichloromethane to establish the pyridine N-oxide functionality
within opicapone preceded methyl ether cleavage through the
use of aluminum trichloride in warm pyridine to furnish
opicapone (X) in 53% yield for the two-step sequence.
The preparation of acid chloride 119 involved the nitration
of commercially available benzoic acid 121 followed by thionyl
chloride-mediated conversion of the resulting nitrobenzoic acid
122 to acid chloride 119. Interestingly, although
the nitration step is low-yielding and involves nitric acid, the
authors report an operationally simple isolation method that
has been exemplified on multiple kilogram scale. No yield was
reported for the conversion of 122 to 119.
オピカポン 上流と下流の製品情報
原材料
準備製品
オピカポン 生産企業
Global( 104)Suppliers
- 923287-50-7
- Opicapone
- Opicapone (BIA 9-1067)
- 5-[3-(2,5-Dichloro-4,6-dimethyl-1-oxido-3-pyridinyl)-1,2,4-oxadiazol-5-yl]-3-nitro-1,2-benzenediol
- BIA 9-1067
- 1,2-Benzenediol, 5-[3-(2,5-dichloro-4,6-dimethyl-1-oxido-3-pyridinyl)-1,2,4-oxadiazol-5-yl]-3-nitro-
- 2,5-dichloro-3-(5-(3,4-dihydroxy-5-nitrophenyl)-1,2,4-oxadiazol-3-yl)-4,6-dimethylpyridine 1-oxide
- CS-1268
- 100G,500G,1KG
- Opicapone-d6
- Inhibitor,inhibit,COMT,Opicapone
- オピカポン
- オピカポン (JAN)
- 2,5-ジクロロ-3-[5-(3,4-ジヒドロキシ-5-ニトロフェニル)-1,2,4-オキサジアゾール-3-イル]-4,6-ジメチルピリジン1-オキシド
- 2,5-ジクロロ-3-[5-(3,4-ジヒドロキシ-5-ニトロフェニル)-1,2,4-オキサジアゾール-3-イル]-4,6-ジメチルピリジン-1-イウム-1-オラート
- オンジェンティス錠
- 2,5-ジクロロ-3-[5-(3,4-ジヒドロキシ-5-ニトロフェニル)-1,2,4-オキサジアゾール-3-イル]-4,6-ジメチルピリジン=1-オキシド