ヨノン (α-,β-混合物) 化学特性,用途語,生産方法
外観
無色~黄色、液体
定義
本品は、次の化学式で表される有機化合物である。
解説
C13H20O(192.30).各種異性体を含む総称であり,代表的なものとして,α-イオノンとβ-イオノンがある.混合物がミカン科Boronia megastigmaから単離されたが,通常,シトラールとアセトンの縮合などにより合成する.
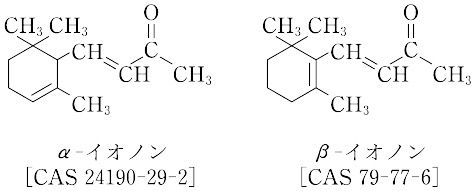
"α-イオノンは沸点127 ℃(1.6 kPa).λmax 229 nm(ε 14300).β-イオノンは沸点134 ℃(1.6 kPa).λmax 294 nm(ε 87000).イロンの代用として香料に用い,またβ-イオノンはビタミンAの合成原料に用いられる.
森北出版「化学辞典(第2版)
化粧品の成分用途
皮膚コンディショニング剤、収れん剤
化学的特性
Light-yellow to colorless liquid; violet
odor.Soluble in
alcohol, ether, mineral oil, and propylene glycol;
insoluble in water and glycerol.
天然物の起源
The α-isomer has been reported in the essential oil of Sphaeranthus inducus L. and in the absolute essence of Acacia farnesiana. The ?-isomer has been reported to be found in raspberry, in the distillate from flowers of Boronia megatisma Nees., and in a few other essences (Fenaroli's Handbook of Flavor Ingredients, 1971). α-Ionone occurs in the essential oils of orange and Ligusticum elatum, in extract of Osmanthus fragrans Lour., in the flavour of tea, and in the essential oil of tangelo (Citrus reticulata Blanco χ C paradisi MacFayden). ?-Ionone is an important constituent of essential oils of Cunila lythrifolia Benth., and Siparuna nicaraguensis Heml. ; it has also been found in tomatoes
使用
Ionone for synthesis. CAS 8013-90-9, molar mass 192.3 g/mol.
製造方法
By chemical synthesis or by condensing citral with acetone to form pseudo-ionone which is then cyclized by acid-type reagents (Bedoukian, 1967).
代謝
Ionones are metabolized mainly by oxidation of the ring system at the carbon atom alpha to the ring double bond and by reduction of the carbonyl group (Williams, 1959). On administration to dogs α-ionone is hydroxylated in the ring at the carbon atom which is alpha to the ring double bond to yield 5-hydroxy-a-ionone (Prelog, Wursch & Meier. 1951). Rabbits dosed orally with /Monone excreted in the urine unchanged ?-ionone, 3-oxo-?-ionone, 3-oxo-?- ionol, dihydro-3-oxo-?-ionol and 3-hydroxy-?-ionol. Excretion products were isolated as 2,4-dinitrophenyl: hydrazone derivatives and as p-nitrobenzoate derivatives. The glucuronides of 3-oxo-?-ionol and dihydro-3- oxo-j?-ionol were also detected in the urine
ヨノン (α-,β-混合物) 上流と下流の製品情報
原材料
準備製品