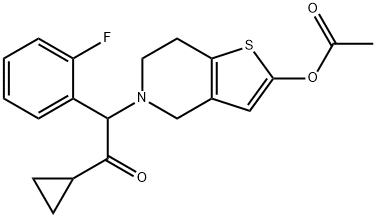
프라수그렐 C화학적 특성, 용도, 생산
개요
Prasugrel is a third-generation thienopyridine that has been developed
and launched for the prevention of atherothrombotic events in patients
with ACS or following PCI. While the second-generation agent clopidogrel
was an improvement over the first-generation ticlopidine, which suffered
from gastrointestinal adverse effects and the risk of neutropenia with
prolonged use, its delayed onset of action and considerable interpatient
variability prompted the search for the next-generation thienopyridine.
The mechanism of action of these platelet inhibitors involves initial biological activation to a sulfhydryl metabolite that irreversibly binds to the
P2γ
12 receptor on platelets via disulfide formation, thereby preventing platelet activation and aggregation by the endogenous agonist adenosine
diphosphate (ADP). The advantage of prasugrel over its predecessors is its
more efficient and consistent absorption and rapid conversion to its active
metabolite. Co-administration of thienopyridines with acetylsalicylic acid
(aspirin), an inhibitor of the synthesis of the platelet aggregation mediator
thromboxane A2, is an effective antiplatelet strategy and joins antagonists
of glycoprotein IIb/IIIa, which target the final step in platelet aggregation,
in the medical arsenal combating atherothrombotic events.
용도
Prasugrel is a platelet inhibitor that reduces aggregation of platelets by being a P2Y12(ADP receptor) inhibitor.
Clinical Use
Prasugrel is a platelet inhibitor developed by Daiichi Sankyo Co.
and is marketed in the United States in cooperation with Eli Lilly and Company for acute coronary syndromes planned for percutaneous
coronary intervention (PCI). Prasugrel was approved for
use in Europe in February 2009, and is currently available in the
UK. In the U.S. prasugrel is also approved for the reduction of
thrombotic cardiovascular events, including stent thrombosis, in
patients with acute coronary syndrome who are to be managed
with PCI. Prasugrel is a member of the thienopyridine class of
ADP receptor inhibitors, and irreversibly binds to P2Y12 receptors.
부작용
In addition to the hemorrhagic side effect, other serious adverse events included AF, bradycardia, leucopenia, severe thrombocytopenia, angiodema, anemia, and abnormal hepatic function with hypertension, headache, back pain, dyspnea, nausea, dizziness, and diarrhea as less severe complaints. Prasugrel is contraindicated in patients with active pathological bleeding, such as peptic ulcers or intracranial hemorrhage, and in patients with a history of prior transient ischemic attack or stroke. In addition, in patients 75 years old, <60 kg, or likely to undergo urgent coronary artery bypass graft surgery, the risk may not outweigh the benefit. When possible, prasugrel treatment should be discontinued at least 7 days prior to any surgery. While warfarin and non-steroidal antiinflammatory drugs (NSAIDS) may increase the risk of bleeding with coadministration of prasugrel, no drug interactions are anticipated with concomitant use of drugs that are inducers or inhibitors of the cytochrome P450 enzymes. Prasugrel may also be administered with aspirin (75-325 mg per day), heparin, GP IIb/IIIa inhibitors, statins, digoxin, and drugs that elevate gastric pH, including PPIs and H2 blockers.
프라수그렐 준비 용품 및 원자재
원자재
Ethanone, 1-cyclopropyl-2-(2-fluorophenyl)-2-[(methylsulfonyl)oxy]-
2-Bromo-2-(2-fluorophenyl)-1-cyclopropylethanone
5-[2-Cyclopropyl-1-(2-fluorophenyl)-2-oxoethyl]-4,5,6,7-tetrahydrothieno[3,2-c]pyridin-2(3H)-one
5,6,7,7a-Tetrahydrothieno[3,2-c]pyridin-2(4H)-one 4-methylbenzenesulfonate
THIENO[3,2-C]PYRIDIN-2(3H)-ONE, 4,5,6,7-TETRAHYDRO-, HYDROCHLORIDE (1:1)
5-[2-Cyclopropyl-1-(2-fluorophenyl)-2-oxoethyl]-5,6,7,7a-tetrahydrothieno[3,2-c]pyridin-2(4H)-one
Desacetyl 2-O-tert-ButyldiMethylsilyl Prasugrel
4,5,6,7-Tetrahydrothieno[3,2-c]pyridin-2(3H)-one 4-methylbenzenesulfonate
Prasugrel (Maleic acid)
아세틸 염화물
무수초산
준비 용품