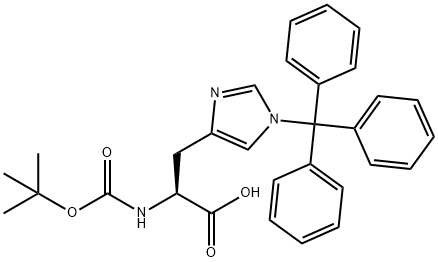
Boc-His(Trt)-OH
|
|
Boc-His(Trt)-OH 속성
- 녹는점
- ~130 °C (dec.)
- 알파
- 12.5 º (C=1% IN MEOH)
- 끓는 점
- 667.5±55.0 °C(Predicted)
- 밀도
- 1.16±0.1 g/cm3(Predicted)
- 저장 조건
- Keep in dark place,Sealed in dry,Room Temperature
- 용해도
- DMSO(약간 용해됨), 메탄올(약간 용해됨)
- 물리적 상태
- 고체
- 물리적 상태
- 단단한 모양
- 산도 계수 (pKa)
- 3.17±0.10(Predicted)
- 색상
- 흰색에서 황백색까지
- optical activity
- [α]20/D +12.5±1.0°, c = 1% in methanol
- BRN
- 732035
- InChIKey
- OYXZPXVCRAAKCM-SANMLTNESA-N
- SMILES
- C(O)(=O)[C@H](CC1N=CN(C(C2=CC=CC=C2)(C2=CC=CC=C2)C2=CC=CC=C2)C=1)NC(OC(C)(C)C)=O
- CAS 데이터베이스
- 32926-43-5(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| WGK 독일 | 3 | ||
|---|---|---|---|
| HS 번호 | 29224999 |
| 그림문자(GHS): |

|
|||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 신호 어: | Warning | |||||||||||||||||||||
| 유해·위험 문구: |
|
|||||||||||||||||||||
| 예방조치문구: |
|
Boc-His(Trt)-OH C화학적 특성, 용도, 생산
화학적 성질
White to off white powder용도
Boc-His(Trt)-OH is a histidine derivative. Its a protected histidine derivative for only the critical coupling step. After coupling the Boc- and Trt-groups are cleaved simultaneously by TFA.용도
Boc-His(Trt)-OH was used in the study to prepare and study structure-activity relationship of amino acid amides as selective inhibitors for dipeptidyl peptidases.주요 응용
N-Boc-N'-Trityl-L-histidine (Boc-His(Trt)-OH) can be used as an intermediate in biochemistry and medicinal chemistry and can be used in the synthesis of peptide drug molecules, vitamins, and molecules with specific biologically active functions. In organic synthesis transformation, the carboxyl group in N-Boc-N'-trityl-L-histidine can undergo corresponding condensation reactions with amine compounds to obtain the corresponding amide products; in addition, in dithionyl chloride Under the action of N-Boc-N'-trityl-L-histidine, intramolecular dehydration cyclization reaction can be performed to obtain cyclic amide derivatives.제조 방법
The benzyl 2-tert-butoxycarbonylamino-3-(1-trimethylimidazol-4-yl)propionate was dissolved in methanol (20 mL), and potassium hydroxide was added to the reaction system. The reaction system was stirred for 1 h after THF was added to the reaction system. The solvent is removed under vacuum depressurization, and the mixture is then extracted with water and Et2O. Separate the two phases and acidify the aqueous layer with dilute HCl, extract it with Et2O, combine all the ether organic layer and wash it with water and saline, dry the organic layer with anhydrous magnesium sulfate, filter to remove the solvent, and concentrate the filtrate under vacuum under reduced pressure to obtain Boc-His(Trt)-OH.reaction suitability
Reaction type: Boc solid-phase peptide synthesisBoc-His(Trt)-OH 준비 용품 및 원자재
원자재
준비 용품
Boc-His(Trt)-OH 공급 업체
글로벌( 218)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Sichuan HongRi Pharma-Tech Co.,Ltd | +86-028-64841719 +8618980456393 |
daisy@enlaibio.com | China | 1369 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29797 | 60 |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-66670886 |
info@dakenam.com | China | 15371 | 58 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 |
fandachem@gmail.com | China | 9352 | 55 |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 |
ivan@atkchemical.com | China | 32480 | 60 |
| AB PharmaTech,LLC | 323-480-4688 |
United States | 989 | 55 | |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| Xiamen AmoyChem Co., Ltd | +86-592-6051114 +8618959220845 |
sales@amoychem.com | China | 6387 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| Sichuan Jiaying Lai Technology Co.,LTD | 18716376478 |
ycling@enlaibio.com | CHINA | 302 | 58 |
Boc-His(Trt)-OH 관련 검색:
파라-톨룰일산 L-(-) 히스티딘 트리틸클로라이드 펜옥심틸 페니실린 톨루엔 1-(트리페닐메틸)이미다졸
N-Carbobenzyloxyglycine
Boc-Aib-OH
Trityl candesartan cilexetil
N-Boc-L-Histidine
N-Boc-N'-Cbz-L-lysine
N-alpha-t-Butyloxycarbonyl-N-epsilon-benzyloxycarbonyl-L-lysinol
Boc-D-Ser(Bzl)-OH
N-BOC-O-Benzyl-L-serine
BOC-GLY-LEU-OH
H-HIS(BZL)-OH
BOC-HIS(BZL)-OH
(-)-2-[METHYLAMINO]-1-PHENYLPROPANE