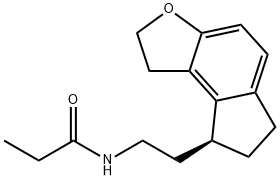
Ramelteon
|
|
Ramelteon 속성
- 녹는점
- 113-1150C
- 알파
- D20 -57.8° (c = 1.004 in chloroform)
- 끓는 점
- 455.3±24.0 °C(Predicted)
- 밀도
- 1.119±0.06 g/cm3(Predicted)
- 인화점
- 2℃
- 저장 조건
- Sealed in dry,Store in freezer, under -20°C
- 용해도
- 디메틸설폭사이드, 에탄올, 메탄올에 용해됨
- 물리적 상태
- 고체
- 물리적 상태
- 단단한 모양
- 산도 계수 (pKa)
- 16.37±0.46(Predicted)
- 색상
- 수정 같은
- InChI
- InChI=1S/C16H21NO2/c1-2-15(18)17-9-7-12-4-3-11-5-6-14-13(16(11)12)8-10-19-14/h5-6,12H,2-4,7-10H2,1H3,(H,17,18)/t12-/m0/s1
- InChIKey
- YLXDSYKOBKBWJQ-LBPRGKRZSA-N
- SMILES
- C(NCC[C@H]1C2=C3CCOC3=CC=C2CC1)(=O)CC
- CAS 데이터베이스
- 196597-26-9(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | F,Xn | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 11-20/21/22-36 | ||
| 안전지침서 | 16-36/37 | ||
| 유엔번호(UN No.) | UN 1648 3 / PGII | ||
| WGK 독일 | 2 | ||
| HS 번호 | 2932.99.7000 | ||
| 유해 물질 데이터 | 196597-26-9(Hazardous Substances Data) |
Ramelteon C화학적 특성, 용도, 생산
개요
Ramelteon, also known as N-[2-[(8S)-1,6,7,8-Tetrahydro-2H-indeno[5,4-b]furan-8-yl]ethyl]propanamide, is a melatonin agonist developed by Takeda Pharmaceuticals, Inc. It was approved by the FDA for marketing in the United States in September 2005 and is marketed under the name Rozerem. It is used to treat difficult-to-sleep and short-term insomnia. Ramelteon is effective for both chronic and short-term insomnia. Unlike most treatments of insomnia that target the GABA (g-aminobutyric acid) receptor complex, ramelteon is an agonist of the melatonin receptor. In particular, it has high selectivity for the MT1 and MT2 subtypes, which have been implicated in the maintenance of circadian rhythms, over the MT3 receptor responsible for other melatonin functions. Its lack of affinity for not only the GABA receptor complex but also neurotransmitter, dopaminerigic, opiate, and benzodiazepine receptors suggests an improved safety profile devoid of the abuse potential of the hypnotic drugs that target these receptors. As such, ramelteon is not a scheduled drug.화학적 성질
Crystalline Solid용도
Ramelteon is a selective melatonin receptor agonist of MT1 and MT2 approved for the treatment of insomnia (trouble in sleeping). It acts as a sedative and hypnotic agent. Ramelteon is the only prescription sleep aid not designated as a Schedule IV controlled substance.일반 설명
The melatonin molecule was modified mainly by replacing the nitrogen of the indole ring with a carbon to give an indane ring and by incorporating 5-methoxyl group in the indole ring into a more rigid furan ring. The selectivity of the resulting ramelteon for MT1 receptor is eight times more than that of MT2 receptor. Unlike melatonin, it is more effective in initiating sleep (MT1 activity) rather than to readjust the circadian rhythm (MT2 activity). It appears to be distinctly more efficacious than melatonin but less efficacious than benzodiazepines as a hypnotic. Importantly, this drug has no addiction liability (it is not a controlled substance). As a result, it has recently been approved for the treatment of insomnia.Synthesis
Vilsmeier-Haack reaction on benzofuran 112 provided aldehyde 113 (100%), which was converted to olefin 114 (88%) by Horner-Emmons reaction with triethylphosphonoacetate, and was followed by hydrogenation of the olefin to give ester 115 (100%). In order to avoid the cyclization of the acid chloride intermediate into the wrong position, the benzene ring was protected by bromination. Both bromination and hydrolysis of the ester is accomplished in a single pot to give acid 116. Thus the ester is brominated with bromine in sodium acetate and acetic acid at 0°C and RT for several hours followed by quenching of remaining bromide by sodium thiosulfate. The resulting acidic solution was taken up in acetonitrile and refluxed for 2hr to provide the acid 116 in 73% yield. The conversion of the acid to acid chloride was done by reacting with thionyl chloride in odichlorobenzene at 40°C for 30 to 40 min after which the reaction was cooled to 0°C . Aluminum trichloride was added and the reaction mixture was stirred at 0°C for 30 min to deliver cyclized ketone 117 in 92% yield. After completion of the cyclization, the bromines are removed by hydrogenation (86%) and resulting ketone 118 was then reacted under Horner-Emmons condition with diethyl cyano phosphonate to give vinyl nitrile 119 in 84% yield. Selective reduction of the nitrile was accomplished by hydrogenation under basic condition (sodium hydroxide in toluene) in the presence of the activated cobalt at 25-50°C for 6.5 hr. The amine was recovered as hydrochloride salt 120 (99% yield) by treating the amine with HCl in methanol. In the next step, the amine salt 120 was taken up in toluene and treated with sodium hydroxide followed by hydrogenation of the mixture with [RuCl(benzene)(R)-BINAP]Cl as catalyst to provide chiral amine 121, after several work up and palladium catalyzed hydrogenations, in 73% overall yield. Final acylation of the amine with propionyl chloride in the presence of aqueous sodium hydroxide in THF at room temperature gave the desired product ramelteon (XVI), after crystallization, in 97% yield.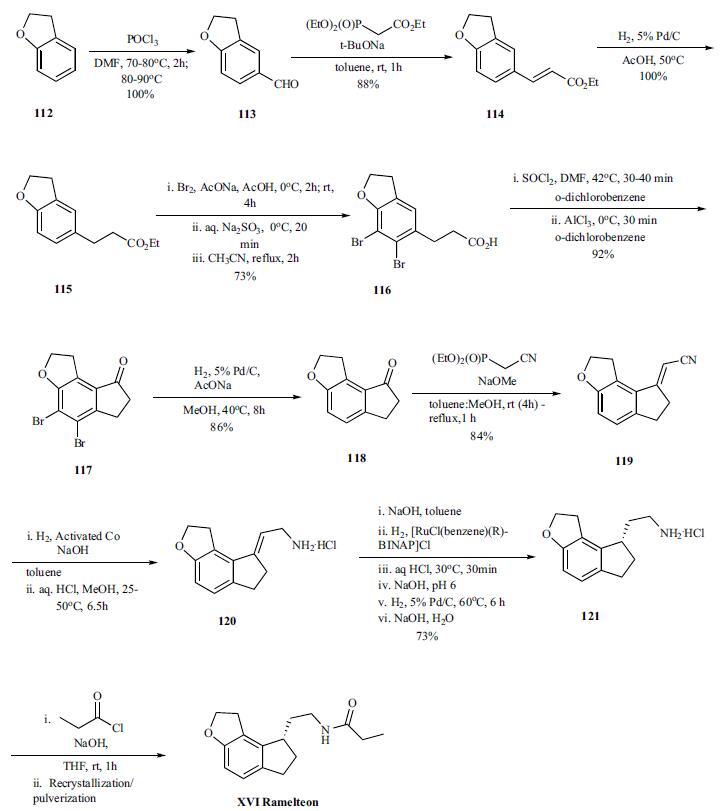
참고 문헌
[1] KATOKOKI. Neurochemical properties of ramelteon (TAK-375), a selective MT1/MT2 receptor agonist.[J]. Neuropharmacology, 2005. DOI:10.1016/j.neuropharm.2004.09.007.[2] MIYAMOTO M. Pharmacology of Ramelteon, a Selective MT1/MT2 Receptor Agonist: A Novel Therapeutic Drug for Sleep Disorders[J]. CNS Neuroscience & Therapeutics, 2009. DOI:10.1111/j.1755-5949.2008.00066.x.
[3] MCGECHANADAM WellingtonKeri. Ramelteon.[J]. CNS drugs, 2005. DOI:10.2165/00023210-200519120-00007.
[4] BORJANANCY L DanielKaren L. Ramelteon for the treatment of insomnia.[J]. Clinical therapeutics, 2006. DOI:10.1016/j.clinthera.2006.10.016.
Ramelteon 준비 용품 및 원자재
원자재
1,2,6,7-Tetrahydro-8H-indeno[5,4-b]furan-8-one
6-메톡시-1-인단온
무수프로피온산
(S)-N-[2-[2,3-Dihydro-6-hydroxy-7-(2-hydroxyethyl)-1H-inden-1-yl]ethyl]propanamide
메탄설포닐 클로라이드
프로피온일 염화물
(S)-2-(2,6,7,8-tetrahydro-1H-indeno[5,4-b]furan-8-yl)acetonitrile
준비 용품
Ramelteon 공급 업체
글로벌( 386)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hong Kong Excellence Biotechnology Co., Ltd. | +86-86-18838029171 +8618126314766 |
ada@sh-teruiop.com | China | 893 | 58 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 |
deasea125996@gmail.com | China | 2503 | 58 |
| Sigma Audley | +86-18336680971 +86-18126314766 |
nova@sh-teruiop.com | China | 525 | 58 |
| Shanghai Aosiris new Material Technology Co., LTD | 86-15139564871 +8615139564871 |
wrjmoon2000@163.com | China | 354 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29797 | 60 |
| Beijing Cooperate Pharmaceutical Co.,Ltd | 010-60279497 |
sales01@cooperate-pharm.com | CHINA | 1811 | 55 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| Shanghai Time Chemicals CO., Ltd. | +86-021-57951555 +8617317452075 |
jack.li@time-chemicals.com | China | 1807 | 55 |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 |
ivan@atkchemical.com | China | 32480 | 60 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |