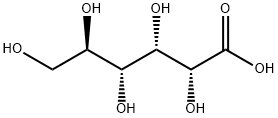
글루콘산
|
|
글루콘산 속성
- 녹는점
- 15 °C
- 끓는 점
- 102 °C
- 알파
- D20 -6.7° (c = 1)
- 밀도
- 1.23
- 굴절률
- 1.4161
- 저장 조건
- Store below +30°C.
- 용해도
- DMSO(약간 용해됨), 메탄올(약간 용해됨), 물
- 물리적 상태
- 결정질 분말 또는 결정
- 산도 계수 (pKa)
- pK (25°) 3.60
- 색상
- 흰색에서 밝은 노란색
- Specific Gravity
- 1.234
- 냄새
- 상업용 50% 수용액. 호박색, 희미한 식초 냄새
- optical activity
- [α]/D +9.0 to 15.5°
- 수용성
- 물에 용해됩니다.
- Merck
- 14,4456
- BRN
- 1726055
- LogP
- -2.116 (est)
- CAS 데이터베이스
- 526-95-4(CAS DataBase Reference)
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xi,C | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 36/37/38-34 | ||
| 안전지침서 | 37/39-26-45-36/37/39 | ||
| 유엔번호(UN No.) | 3265 | ||
| WGK 독일 | - | ||
| RTECS 번호 | LZ5057100 | ||
| TSCA | Yes | ||
| 위험 등급 | 8 | ||
| 포장분류 | III | ||
| HS 번호 | 29181600 | ||
| 유해 물질 데이터 | 526-95-4(Hazardous Substances Data) | ||
| 기존화학 물질 | KE-17664 |
글루콘산 C화학적 특성, 용도, 생산
개요
글루콘산은 부드럽고, 청량한 산미(酸味)를 가지므로, 주류, 식초, 청량음료의 첨가물로 사 용됩니다. 철제품의 방청력이 강해 철제품이 쉽게 녹슬지 않도록 하는데 사용됩니다. 금속표면의 산화물, 스케일등을 금속자체의 부식없이 쉽게 제거하므로 탈청제, 스케일제거 제 등으로 사용됩니다.용도
식품 : 주류, 식초, 청량음료등의 산미료(酸味料), 식품제조 장치 및 용기의 스케일 제거 및 세척제 등.금속 : 세관제, 금속처리제, 방청제, 탈지제.
섬유 : 염색보조제.
순도시험
(1) 염화물 : 이 품목 0.5g을 취하여 염화물시험법에 따라 시험할 때, 그 양은 0.01N 염산 0.5mL에 대응하는 양 이하이어야 한다.
(2) 황산염 : 이 품목 1g을 취하여 황산염시험법에 따라 시험할 때, 그 양은 0.01N 황산 0.5mL에 대응하는 양 이하이어야 한다.
(3) 비소 : 이 품목을 비소시험법에 따라 시험할 때, 그 양은 4.0ppm 이하이어야 한다.
(4) 납 : 이 품목 5.0g을 취하여 원자흡광광도법 또는 유도결합플라즈마발광광도법에 따라 시험할 때, 그 양은 5.0ppm 이하이어야 한다.
(5) 수은 : 이 품목을 수은시험법에 따라 시험할 때, 그 양은 1.0ppm 이하이어야 한다.
(6) 자당 또는 환원당 : 이 품목 1g에 물 10mL 및 묽은염산 2mL를 가하여 2분간 끓이고 식힌 다음 탄산나트륨시액 5mL를 가하여 4분간 방치한 다음 물을 가하여 20mL로 하고 이 액 5mL에 펠링시액 2mL를 가하여 1분간 끓일 때, 즉시 등황~적색의 침전이 생겨서는 아니 된다.
확인시험
(1) 이 품목의 수용액(1→25) 1mL에 염화제이철시액 1방울을 가할 때, 진한 황색을 나타낸다.
(2) 이 품목 1mL에 물 4mL를 가하여 「글루코노델타락톤」의 확인시험 (3)에 따라 시험한다.
정량법
이 품목 1g을 정밀히 달아 물 30mL 및 0.1N 수산화나트륨용액 40mL를 가하여 흔들어 섞고 20분간 방치한 다음 과잉의 알칼리를 0.1N 황산으로 적정한다(지시약 : 페놀프탈레인시액 3방울). 따로 같은 방법으로 공시험을 한다.
0.1N 수산화나트륨용액 1mL = 19.616mg C6H12O7
정의
이 품목은 글루콘산 및 글루코노-δ-락톤의 수용액이다.
포장, 보관
철 및 이온성 금속, 알루미늄 재질은 부식성이 있으므로 스테인레스 316, 304, 프라스틱, 유리재질의 용기사용. 45% 용액은 저온 (4℃)에서 흰색 결정이 석출될 수 있으며, 결정 석출 시 가온하면 쉽게 녹음. 15℃ 이상에서 저장하는 것이 좋음.강열잔류물
이 품목 5g을 취하여 강열잔류물시험법에 따라 시험할 때, 그 양은 0.10% 이하이어야 한다.
개요
Gluconic acid is an organic compound with molecular formula C6H12O7 and condensed structural formula HOCH2(CHOH)4COOH. It is one of the 16 stereoisomers of 2,3,4,5,6-penta hydroxy hexanoic acid.In aqueous solution at neutral pH, gluconic acid forms the gluconate ion. The salts of gluconic acid are known as "gluconates". Gluconic acid, gluconate salts, and gluconate esters occur widely in nature because such species arise from the oxidation of glucose. Some drugs are injected in the form of gluconates.
화학적 성질
d-Gluconic acid is an acid sugar composed of white crystals with a milk-acidic taste. In aqueous solutions, it is in equilibrium with gamma- and delta-gluconolactones. It is prepared by enzymatic oxidation of glucose and strains of the microorganisms used to supply the enzyme action are nonpathogenic and nontoxicogenic to man or other animals. This substance is used as a component of bottle rinsing formulations, at levels not to exceed good manufacturing practice.An FDA letter to a trade association revoking affirmation of general recognition of safety in dietary supplements was dated April 9, 1970.
물리적 성질
The chemical structure of gluconic acid consists of a six-carbon chain with five hydroxyl groups terminating in a carboxylic acid group. In aqueous solution, gluconic acid exists in equilibrium with the cyclic ester glucono delta-lactone.출처
Gluconic acid occurs naturally in fruit, honey, kombucha tea, and wine. As a food additive ( E574 ), it is an acidity regulator. It is also used in cleaning products where it dissolves mineral deposits especially in alkaline solution. The gluconate anion chelates Ca2+,Fe2+, Al3+, and other metals. In 1929 Horace Terhune Herrick developed a process for producing the salt by fermentation.Calcium gluconate, in the form of a gel, is used to treat burns from hydrofluoric acid; calcium gluconate injections may be used for more severe cases to avoid necrosis of deep tissues. Quinine gluconate is a salt between gluconic acid and quinine, which is used for intramuscular injection in the treatment of malaria. Zinc gluconate injections are used to neuter male dogs. Iron gluconate injections have been proposed in the past to treat anemia.
용도
Gluconic Acid is an acidulant that is a mild organic acid which is the hydrolyzed form of glucono-delta-lactone. it is prepared by the fermentation of dextrose, whereby the physiological d-form is produced. it is soluble in water with a solubility of 100 g/100 ml at 20°c. it has a mild taste and at 1% has a ph of 2.8. it functions as an antioxidant and enhances the function of other antioxidants. in beverages, syrups, and wine, it can eliminate calcium turbidities. it is used as a leavening component in cake mixes, and as an acid component in dry-mix desserts and dry beverage mixes.정의
gluconic acid: An opticallyactive hydroxycarboxylic acid,CH2(OH)(CHOH)4COOH. It is the carboxylicacid corresponding to the aldosesugar glucose, and can be madeby the action of certain moulds.글루콘산 준비 용품 및 원자재
원자재
준비 용품
글루콘산 공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Yancheng Green Chemicals Co.,Ltd | +undefined-86-25-86655873 |
info@royal-chem.com | China | 114 | 58 |
| Gansu Jinyuantai New Material Co., Ltd. | +86-025-86618065 +86-13914714331 |
sales@jinyuantaichem.com | China | 73 | 58 |
| Ningxia Jinhua Chemical Co.,Ltd | 025-52279164 |
info@nxjhchem.com | China | 79 | 58 |
| Changde Changlian Chemical Co., Ltd. | +86-25-86736270 +86-15905172087 |
brian.he@royal-chem.com | China | 103 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12453 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21695 | 55 |
| Nanjing ChemLin Chemical Industry Co., Ltd. | 025-83697070 |
product@chemlin.com.cn | CHINA | 3012 | 60 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 |
info@tnjchem.com | China | 2989 | 55 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |