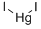요오드화수은(II)(적색) C화학적 특성, 용도, 생산
화학적 성질
Red Solid
용도
Mercury(II) iodide is is used for preparation of Nessler's reagent, used for detection of presence of ammonia. It is a semiconductor material, used in some x-ray and gamma ray detection and imaging devices operating at room temperatures. In veterinary medicine, it is used in blister ointments in exostoses, bursal enlargement, etc.
일반 설명
A scarlet-red odorless tasteless powder. Sensitive to light. Insoluble in water and sinks in water. At elevated temperatures turns yellow but turns back to red upon cooling. Poison.
공기와 물의 반응
Insoluble in water.
반응 프로필
MERCURY(II) IODIDE is a mild reducing agent. Reacts with sodium azide to form mercury(II) azide, which is shock, friction, and heat sensitive. Incompatible with acetylene, ammonia, chlorine dioxide, azides, chlorine trifluoride, calcium (because of amalgam formation), sodium carbide, lithium, rubidium, copper .
위험도
Highly toxic by ingestion, inhalation, and
skin absorption; strong irritant.
건강위험
All forms of exposure to MERCURY(II) IODIDE are hazardous. Acute systemic mercurialism may be fatal within a few minutes; death by uremic poisoning is usually delayed 5-12 days. Acute poisoning has resulted from inhaling dust concentrations of 1.2-8.5 mg/m 3 of air; symptoms include tightness and pain in chest, coughing, and difficulty in breathing. Ingestion causes necrosis, pain, vomiting, and severe purging. Contact with eyes causes ulceration of conjunctiv a and cornea. Contact with skin causes irritation and possible dermatitis; systemic poisoning can occur by absorption through skin.
화재위험
Special Hazards of Combustion Products: Fumes from fire may contain toxic mercury vapor.
Safety Profile
A poison. When heated
to decomposition it emits very toxic fumes
of Hg and I-. See also MERCURY(I1)
IODIDE.
잠재적 노출
Mercuric iodide is used in medicine
and in analytical chemistry.
Purification Methods
Crystallise it from MeOH or EtOH and wash it repeatedly with distilled water (solubility is 0.006% at ~25o). It has also been mixed thoroughly with excess 0.001M iodine solution, filtered, washed with cold distilled water, rinsed with EtOH and Et2O, and dried in air. It changes colour reversibly to yellow at ~130o. [Friend Nature 109 341 1922.] POISONOUS.
비 호환성
Violent reaction with active metals;
potassium, sodium, acids, chlorine trifluoride. Inorganic
mercury compounds are incompatible with acetylene,
ammonia, chlorine dioxide; azides, calcium (amalgam formation), sodium carbide, lithium, rubidium, copper.
Mercury iodide is a mild reducing agent. Keep away from
oxidizers. Reacts with sodium azide to form mercury(II)
azide, which is shock-, friction-, and heat-sensitive.
Incompatible with acetylene, ammonia, chlorine dioxide,
azides, chlorine trifluoride, calcium (because of amalgam
formation), sodium carbide, lithium, rubidium, copper
(NIOSH, 1997)
요오드화수은(II)(적색) 준비 용품 및 원자재
원자재
준비 용품