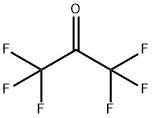
헥사플루오로아세톤 C화학적 특성, 용도, 생산
화학적 성질
Hexafluoroacetone is a colorless, nonflamma ble poison gas. Musty odor. Shipped as a liquefied com pressed gas.
용도
Hexafluoroacetone is a protecting and activatng reagent used in the synthesis of (S)-isoserine from (S)-malic acid. It is also an intermediate used to prepare hexafluorocarbinols as liver X receptor-α agonists.
정의
ChEBI: A ketone that is acetone in which all the methyl hydrogens are replaced by fluoro groups.
일반 설명
Hexafluoroacetone is a colorless, toxic, and highly reactive gas. At ambient temperatures, Hexafluoroacetone is likely to generate a considerable amount of vapor. Hexafluoroacetone is an irritant to skin, eyes and mucous membranes and is toxic by ingestion, skin absorption, and inhalation. When heated to high temperatures Hexafluoroacetone emits toxic fluoride fumes. Prolonged exposure of the container to fire or intense heat may cause Hexafluoroacetone to violently rupture and rocket. Hexafluoroacetone is used in the production of other chemicals.
공기와 물의 반응
Hygroscopic (i.e., absorbs moisture from the air); reacts with moisture to form a highly acidic sesquihydrate. .
반응 프로필
Hexafluoroacetone is incompatible with the following: Water, acids [Note: Hygroscopic (i.e., absorbs moisture from the air); reacts with moisture to form a highly acidic sesquihydrate.] .
위험도
Toxic by inhalation and skin absorption.
Reacts vigorously with water and other substances,
releasing considerable heat. Nonflammable.
건강위험
TOXIC; may be fatal if inhaled, ingested or absorbed through skin. Vapors are extremely irritating and corrosive. Contact with gas or liquefied gas may cause burns, severe injury and/or frostbite. Fire will produce irritating, corrosive and/or toxic gases. Runoff from fire control may cause pollution.
화재위험
Some may burn but none ignite readily. Vapors from liquefied gas are initially heavier than air and spread along ground. Some of these materials may react violently with water. Cylinders exposed to fire may vent and release toxic and/or corrosive gas through pressure relief devices. Containers may explode when heated. Ruptured cylinders may rocket.
잠재적 노출
Hexafluoroacetone is used as a chemi cal intermediate. A gas at room temperature, it forms vari ous hydrates with water which are used as solvents for
resins and polymers. Other derivatives are used to make
water repellent coatings for textiles and also to produce
polymers.
운송 방법
UN2420 Hexafluoroacetone, Hazard Class: 2.3;
Labels: 2.3-Poisonous gas, 8-Corrosive material, Inhalation
Hazard Zone B. Cylinders must be transported in a secure
upright position, in a well-ventilated truck. Protect cylinder
and labels from physical damage. The owner of the com pressed gas cylinder is the only entity allowed by federal
law (49CFR) to transport and refill them. It is a violation
of transportation regulations to refill compressed gas cylin ders without the express written permission of the owner.
Purification Methods
Dehydrate hexafluoroacetone by passing the vapours over P2O5. Ethylene is removed by passing the dried vapours through a tube containing Pyrex glass wool moistened with conc H2SO4. Further purification is by low temperature distillation using Warde-Le Roy stills. Store it in the dark at -78o. [Holmes & Kutschke Trans Faraday Soc 58 333 1962, Beilstein 1 IV 3215.]
비 호환성
Reacts with water, oxidizers, strong
acids. Hygroscopic (i.e., absorbs moisture from the air);
reacts with moisture to form a highly acidic sesquihydrate
and considerable heat.
폐기물 처리
Return refillable compressed
gas cylinders to supplier. Nonrefillable cylinders should be
disposed of in accordance with local, state and federal
regulations. Allow remaining gas to vent slowly into
atmosphere in an unconfined area or exhaust hood.
Refillable-type cylinders should be returned to original sup plier with any valve caps and outlet plugs secured and
valve protection caps in place.
헥사플루오로아세톤 준비 용품 및 원자재
원자재
준비 용품