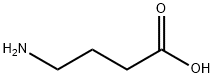
감마-아미노-N-부티르 산
|
|
감마-아미노-N-부티르 산 속성
- 녹는점
- 195 °C (dec.) (lit.)
- 끓는 점
- 248.0±23.0 °C(Predicted)
- 밀도
- 1.2300 (estimate)
- 굴절률
- 1.4650 (estimate)
- 저장 조건
- 2-8°C
- 용해도
- H2O: 1 M at 20 °C, 투명, 무색
- 산도 계수 (pKa)
- 4.031(at 25℃)
- 물리적 상태
- 가루
- 색상
- 흰색에서 거의 흰색
- 냄새
- 프로필렌 글리콜 중 1.00%. 풍미있는 고기
- ?? ??
- 고기가 많은
- 수용성
- 녹는
- Merck
- 14,430
- JECFA Number
- 1771
- BRN
- 906818
- InChIKey
- BTCSSZJGUNDROE-UHFFFAOYSA-N
- LogP
- -3.17
- CAS 데이터베이스
- 56-12-2(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xi,Xn | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 36/37/38-20/21/22 | ||
| 안전지침서 | 26-36 | ||
| WGK 독일 | 2 | ||
| RTECS 번호 | ES6300000 | ||
| 위험 참고 사항 | Irritant | ||
| TSCA | Yes | ||
| HS 번호 | 29224995 |
감마-아미노-N-부티르 산 C화학적 특성, 용도, 생산
개요
4-Aminobutyric acid (GABA) is the chief inhibitory neurotransmitter in the mammalian central nervous system. It plays a role in regulating neuronal excitability throughout the nervous system. In humans, GABA is also directly responsible for the regulation of muscle tone. Although chemically it is an amino acid, GABA is rarely referred to as such in the scientific or medical communities, because the term "amino acid," used without a qualifier, conventionally refers to the alpha amino acids, which GABA is not, nor is it ever incorporated into a protein. In spastic diplegia in humans, GABA absorption becomes impaired by nerves damaged from the condition's upper motor neuron lesion, which leads to hypertonia of the muscles signaled by those nerves that can no longer absorb GABA.화학적 성질
4-Aminobutyric acid is a white flake or needle-like crystal; slightly odorous, deliquescence; easily soluble in water, slightly soluble in hot ethanol, insoluble in cold ethanol, ether and benzene; decomposition point is 202°C; LD50 (rat, abdominal cavity) 5400mg/kg.역사
4-Aminobutyric acid was first synthesized in 1883, and was first known only as a plant and microbe metabolic product. In 1950, however, GABA was discovered to be an integral part of the mammalian central nervous system.용도
4-Aminobutyric acid is an important inhibitory neurotransmitter in the central nervous system, which has good water solubility and thermal stability. It has been confirmed that GABA, as a small molecular weight non protein amino acid, has edible safety and can be used in the production of beverages and other foods. Studies have shown that a certain amount of GABA can improve the body's sleep quality and reduce blood pressure.The foods contain γ-aminobutyric acid (GABA) at an amount that shows immediate effect of suppressing autonomic nerve activity related to blood pressure increase. Reacts with isothiocyanates to produce thioureas which have antifungal activity.제조 방법
The synthesis of 4-aminobutyric acid mainly includes the following: the first is the use of potassium Phthaloyl imine and γ- Chloroprene cyanogen or butyrolactone is used as the raw material of GABA. The final product obtained after violent reaction and hydrolysis is GABA; The second is to use pyrrolidone as the initial raw material, hydrolyze it through calcium hydroxide and ammonium bicarbonate, and finally open its ring to obtain GABA; The third is to use butyric acid and ammonia as raw materials of GABA γ GABA was obtained by light reaction under X-ray conditions; The fourth method is to synthesize GABA with propylamine and formic acid by glow discharge; The fifth is to use methyl bromoacetate and ethylene as raw materials to prepare GABA. Methyl 4-bromobutyrate is obtained through polymerization. Finally, the product after ammonolysis and hydrolysis is GABA. The chemical synthesis methods of GABA have the disadvantages of difficult reaction control and high cost.정의
4-Aminobutyric acid is a gamma-amino acid that is butanoic acid with the amino substituent located at C-4. It has a role as a signalling molecule, a human metabolite, a Saccharomyces cerevisiae metabolite and a neurotransmitter. It is a gamma-amino acid and a monocarboxylic acid. It derives from a butyric acid. It is a conjugate acid of a gamma-aminobutyrate. It is a tautomer of a gamma-aminobutyric acid zwitterion.일반 설명
4-Aminobutyric acid is a chief inhibitory neurotransmitter, which is found in the cerebellum, hypothalamus, thalamus and hippocampus. It is formed via the decarboxylation of L-glutamate catalyzed by the enzyme, glutamic acid decarboxylase(GAD).Mechanism of action
4-Aminobutyric acid (GABA) probably represents the most important inhibitory transmitter of the mammalian CNS. Both types of GABAergic inhibition (pre- and postsynaptic) use the same GABAA receptor subtype, which acts by regulation of the chloride channel of the neuronal membrane. A second GABA receptor type, GABAB, that is a G protein–coupled receptor is not considered to be important in understanding the mechanism of hypnotics. Activation of a GABAA receptor by an agonist increases the inhibitory synaptic response of central neurons to GABA through hyperpolarization. Because many, if not all, central neurons receive some GABAergic input, this leads to a mechanism by which CNS activity can be depressed. For example, if the GABAergic interneurons are activated by an agonist that inhibits the monoaminergic structures of the brainstem, hypnotic activity will be observed. The specific neuronal structures in different brain regions affected by GABAA agonist continues to be better defined.Pharmacology
Drugs that act as allosteric modulators of GABA receptors (known as GABA analogues or GABAergic drugs) or increase the available amount of GABA typically have relaxing, anti-anxiety, and anti-convulsive effects. Many of the substances below are known to cause anterograde amnesia and retrograde amnesia.In general, GABA does not cross the blood–brain barrier, although certain areas of the brain that have no effective blood–brain barrier, such as the periventricular nucleus, can be reached by drugs such as systematically injected GABA. At least one study suggests that orally administered GABA increases the amount of Human Growth Hormone. GABA directly injected to the brain has been reported to have both stimulatory and inhibitory effects on the production of growth hormone, depending on the physiology of the individual.
Source
4-Aminobutyric acid can be found in various foods, including:Tea
Tomatoes
Soybeans
Germinated rice
Some fermented foods, such as tempe and kimchi
Some vegetables, such as broccoli, spinach, and kale
GABA is also widely available as a supplement in either powder or pill form.
신진 대사
GABA transaminase enzyme catalyzes the conversion of 4- aminobutanoic acid and 2-oxoglutarate into succinic semialdehyde and glutamate. Succinic semialdehyde is then oxidized into succinic acid by succinic semialdehyde dehydrogenase and as such enters the citric acid cycle as a usable source of energy.Purification Methods
Crystallise GABA from aqueous EtOH or MeOH/Et2O. Also crystallise it by dissolving it in the least volume of H2O and adding 5-7 volumes of absolute EtOH.감마-아미노-N-부티르 산 준비 용품 및 원자재
원자재
Cation exchange resin,strong acidic styrene
Potassium phthalimide
N-메틸-2-피롤리디논
대두 기름
카프로일클로라이드
2-피롤리디논
클로로부틸로니트릴(4-)
N,N,N-Tri(polyoxypropylene polyoxyethylene)amine
corn syrup
암모늄 비카보네이트
준비 용품
감마-아미노-N-부티르 산 공급 업체
글로벌( 952)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Dayang Chem (Hangzhou) Co.,Ltd. | 571-88938639 +8617705817739 |
info@dycnchem.com | CHINA | 52867 | 58 |
| Xi'an Kono chem co., Ltd., | 029-86107037 13289246953 |
info@konochemical.com | China | 2995 | 58 |
| Sinoway Industrial co., ltd. | 0592-5800732; +8613806035118 |
xie@china-sinoway.com | China | 992 | 58 |
| Baynoe Chem (Suzhou) Co., Ltd. | +86-512 65869404 +86-18934593683 |
sales06@baynoe.com | China | 292 | 58 |
| Suzhou Yuanrui Bio-Tech Co., Ltd | +86-18106132862 +86-18106132862 |
sales02@yuanruibio.com | China | 278 | 58 |
| Leading Chemical and Trading Co.,Ltd | +8615669938129 |
sales@leadingchemical.com | China | 121 | 58 |
| BINBO BIOLOGICAL CO.,LTD | +8618629063126 |
info@binbobiological.com | China | 290 | 58 |
| Jinan Jianfeng Chemical Co., Ltd | 0531-88110457; +8615562555968 |
info@pharmachemm.com | China | 222 | 58 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 |
deasea125996@gmail.com | China | 2503 | 58 |
| Wuhan Haorong Biotechnology Co.,ltd | +8618565342920 |
sales@chembj.net | China | 269 | 58 |