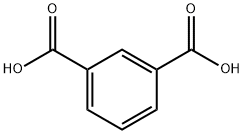
이소프탈산
|
|
이소프탈산 속성
- 녹는점
- 341-343 °C (lit.)
- 끓는 점
- 214.32°C (rough estimate)
- 밀도
- 1,54 g/cm3
- 증기압
- 0Pa at 25℃
- 굴절률
- 1.5100 (estimate)
- 저장 조건
- Sealed in dry,Room Temperature
- 용해도
- 0.12g/L
- 산도 계수 (pKa)
- 3.54(at 25℃)
- 물리적 상태
- 결정성 분말
- 색상
- 흰색에서 황백색까지
- 수소이온지수(pH)
- 3.33(1 mM solution);2.76(10 mM solution);2.24(100 mM solution)
- 수용성
- 0.01 g/100 mL (25 ºC)
- Merck
- 14,5197
- BRN
- 1909332
- Dielectric constant
- 1.4(Ambient)
- 안정성
- 안정적인. 강한 산화제, 강한 염기와 호환되지 않습니다.
- InChIKey
- QQVIHTHCMHWDBS-UHFFFAOYSA-N
- LogP
- 1.66 at 25℃
- CAS 데이터베이스
- 121-91-5(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 36/37/38 | ||
| 안전지침서 | 24/25-36-26 | ||
| WGK 독일 | 2 | ||
| RTECS 번호 | NT2007000 | ||
| 자연 발화 온도 | 1198 °F | ||
| TSCA | Yes | ||
| HS 번호 | 29173980 | ||
| 유해 물질 데이터 | 121-91-5(Hazardous Substances Data) | ||
| 기존화학 물질 | KE-02189 |
이소프탈산 C화학적 특성, 용도, 생산
개요
Isophthalic acid is an organic compound with the formula C6H4(CO2H)2. This colourless solid is an isomer of phthalic acid and terephthalic acid. These aromatic dicarboxylic acids are used as precursors (in form of acyl chlorides) to commercially important polymers, e.g. the fire-resistant material Nomex. Mixed with terephthalic acid, iso phthalic acid is used in the production of resins for drink bottles. The high-performance polymer poly benzimidazole is produced from iso phthalic acid.화학적 성질
Isophthalic acid is a white crystalline powder or needle-like crystals and it’s an isomer of phthalic acid and terephthalic acid.It is insoluble in cold water but soluble in oxygenated solvents and alcohol. It is combustible and finely dispersed particles will form explosive mixtures in air.
Isophthalic Acid (PIA) is mainly used in the production of bottle PET, also used in the production of alkyd resin, polyester resin, also used in the production of photosensitive materials, pharmaceutical intermediates and so on.
One of the largest applications for PIA is in unsaturated polyester resins for high-quality gel coats. The hardness, stain and detergent resistance characteristics of PIA are ideal for polyester solid-surface countertops that are an inexpensive alternative to acrylics.
용도
Purified Isophthalic Acid (PIA) is mainly used as intermediate for high performance UPR, resins for coatings, high solids paints, gel coats, modifier of PET for bottles. Product Data Sheet제조 방법
Iso phthalic acid is produced on the billion kilogram per year scale by oxidizing meta-xylene using oxygen . The process employs a cobalt-manganese catalyst. In the laboratory, chromic acid can be used as the oxidant. It also arises by fusing potassium meta-sulpho benzoate , or meta - brom benzoate with potassium formate (terephthalic acid is also formed in the last case).The barium salt (as its hexa hydrate) is very soluble (a distinction between phthalic and terephthalic acids). Uvitic acid, 5- methylisophthalic acid, is obtained by oxidizing mesitylene or by condensing pyroracemic acid with baryta water.
정의
ChEBI: A benzenedicarboxylic acid that is benzene substituted by carboxy groups at position 1 and 3. One of three possible isomers of benzenedicarboxylic acid, the others being phthalic and terephthalic acids.일반 설명
White solid with a slight unpleasant odor. Sinks in water.공기와 물의 반응
Dust forms explosive mixture in air [USCG, 1999].건강위험
May cause slight to moderate irritation of eyes, skin, and mucous membranes on prolonged contact. Ingestion may cause gastrointestinal irritation.화재위험
Behavior in Fire: Dust forms explosive mixture in air.Purification Methods
Crystallise the acid from aqueous EtOH. [Beilstein 9 IV 3292.]이소프탈산 준비 용품 및 원자재
원자재
코발트(II) 아세테이트
이소프탈로니트릴
3-자일렌
망간(II) 아세테이트 테트라하이드레이트
과망간산칼륨
아세트산(빙초산)
Methyl 3-bromobenzoate
3-Carboxybenzaldehyde
준비 용품
5-Amino-2,4,6-triiodo-N-methylisophthalamic Acid
2,4,5-트리플루오로벤조산
DIETHYL ISOPHTHALATE
2-Iodoxybenzoic acid
이소프탈로니트릴
2,6-Dicyano-4-Nitroaniline
5-AMINO-N-METHYLISOPHTHALAMIC ACID
이소프탈산디메틸
5-니트로이소프탈릭 산
polyester fibre coloring modifier
Sodium dimethyl 5-sulphonatoisophthalate
DIMETHYL 5-BROMOISOPHTHALATE
Biphenyl-3,4′,5-tricarboxylic acid
cis-1,3-cyclohexanedicarboxylic acid
이소프탈산 공급 업체
글로벌( 622)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Wuhan Boyuan Import & Export Co., LTD | +8615175982296 |
Mike@whby-chem.com | China | 974 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12453 | 58 |
| Hebei Yanxi Chemical Co., Ltd. | +8617531190177 |
peter@yan-xi.com | China | 5993 | 58 |
| Hebei Dangtong Import and export Co LTD | +8615632927689 |
admin@hbdangtong.com | China | 991 | 58 |
| Shaanxi Haibo Biotechnology Co., Ltd | +undefined18602966907 |
qinhe02@xaltbio.com | China | 1000 | 58 |
| Hebei Jingbo New Material Technology Co., Ltd | +8619931165850 |
hbjbtech@163.com | China | 1000 | 58 |
| Shanghai Affida new material science and technology center | +undefined15081010295 |
2691956269@qq.com | China | 359 | 58 |
| hebei hongtan Biotechnology Co., Ltd | +86-86-1913198-3935 +8617331935328 |
sales03@chemcn.cn | China | 951 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29797 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21695 | 55 |