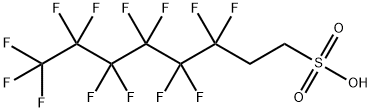
1H,1H,2H,2H-Perfluorooctanesulfonic acid
|
|
1H,1H,2H,2H-Perfluorooctanesulfonic acid 속성
- 밀도
- 1,25 g/cm3
- 증기압
- 1.96Pa at 20℃
- 용해도
- Acetone: Slightly soluble,Chloroform: Slightly soluble,DMSO: Slightly soluble,Methanol: Slightly soluble
- 물리적 상태
- A low-melting solid
- 산도 계수 (pKa)
- 1.31±0.50(Predicted)
- 수용성
- 658g/L at 20℃
- InChI
- InChI=1S/C8H5F13O3S/c9-3(10,1-2-25(22,23)24)4(11,12)5(13,14)6(15,16)7(17,18)8(19,20)21/h1-2H2,(H,22,23,24)
- InChIKey
- VIONGDJUYAYOPU-UHFFFAOYSA-N
- SMILES
- C(S(O)(=O)=O)CC(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | C,Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 34 | ||
| 안전지침서 | 26-36/37/39-45 | ||
| 유엔번호(UN No.) | 3265 | ||
| 위험 참고 사항 | Corrosive | ||
| 위험 등급 | IRRITANT, CORROSIVE | ||
| HS 번호 | 2904990090 | ||
| 기존화학 물질 | 2015-3-6285 |
1H,1H,2H,2H-Perfluorooctanesulfonic acid C화학적 특성, 용도, 생산
개요
1H,1H,2H,2H-Perfluorooctanesulfonic acid is an efficient fluorine-containing surfactant without perfluorooctane sulfonic acid, which has the characteristics of high temperature resistance, oxidant resistance, electrolytic stability, etc. It can reduce the concentration of chromic acid in plating workshop and inhibit the precipitation of chromic acid and chromium mist.화학적 성질
White to Brown Solid.용도
1H,1H,2H,2H-Perfluorooctanesulfonic acid is an aliphatic compound for fluorinated surfactant synthesis.주요 응용
1H,1H,2H,2H-Perfluorooctanesulfonic acid can functionalize gallium nitride (GaN) to tune the optical properties, which can potentially be used in chemical sensor based applications. It can modify the surface characteristics of copper substrates that find usage in printed circuit boards as copper foils. It can also be coated on the indium tin oxide substrate, which may be utilized in organic light emitting diodes (OLEDs) and organic photovoltaics (OPVs).일반 설명
1H,1H,2H,2H-Perfluorooctanesulfonic acid is a fluoroalkyl phosphonic acid that contains 8 fluorinated carbon atoms. It forms a self-assembled layer on the substrate due to the adhesion of the phosphonates. The surface adhesion is a component of the acid base linkage that is enhanced by the hydroxyl groups.Toxicology
Toxicological studies of 1H,1H,2H,2H-Perfluorooctanesulfonic acid (6:2 FTS) induced cardiovascular abnormalities in zebrafish embryos and larvae showed that the acute toxicity of 6:2 FTS was lower than that of PFOS. 6:2 FTS could affect the development of zebrafish, including increasing the utilization of yolk sac and the length of the pharyngeal pouch, leading to pericardial edema and inhibiting beating of the heart. Then transgenic zebrafish CZ63 and CZ40 were used to further explore its cardiovascular toxicity. 6:2 FTS could induce vascular hyperplasia, inhibit atrial development, and reduce blood flow velocity in larvae at 72 hours postfertilization. In addition, 6:2 FTS enhanced CAT and GSH-Px activities, involved in calcium signalling and myocardial contraction, and affected MAPK, FoxO and p53 signalling pathways[3].저장
2-8°CEnvironmental considerations
Regulatory efforts are underway to limit the use of some Fluorinated additive material, mainly perfluorinated compounds (PFCs), due to concerns over their environmental persistency, bioaccumulation ability, and toxicity. Among the PFCs, perfluorosulfonates (PFSAs) and perfluorocarboxylates (PFCAs) are most widely detected in human and other biological samples, including those in pristine areas such as the Arctic. The longer-chained PFCs are known to be bioaccumulative and have toxic effects on biota. For example, neuroendocrine effects and peroxisome proliferation have been shown to occur. The perfluorinated sulfonamides are neutral compounds and are consequently not as water-soluble as the acids and are more volatile. It has been suggested that they are transformed into perfluorinated sulfonates (PFSAs) and PFCAs in the atmosphere. Since the perfluorinated acids have high water solubilities and low pKa values, they are dissociated at environmentally relevant pH values. They are primarily found in water or bound to particles, sediments, and soils[1-2].1H,1H,2H,2H-Perfluorooctanesulfonic acid 준비 용품 및 원자재
원자재
준비 용품
1H,1H,2H,2H-Perfluorooctanesulfonic acid 공급 업체
글로벌( 122)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Tianjin Huiren Chemtech Co., Ltd | +86-86-22-23707858 +86-86-13672008665 |
huirentech@foxmail.com | China | 169 | 58 |
| Wuhan Bright Chemical Co., Ltd | +86-02785804899 +86-15071395570 |
alice@brightchemical.com.cn | China | 93 | 58 |
| Tangshan Moneide Trading Co., Ltd. | +86-315-8309571 +8615633399667 |
sales@moneidechem.com | China | 568 | 58 |
| WEIFANG OUCHEM TRADE CO.,LTD. | +86-18506360988 +86-18506360988 |
wtaili@ouchem.cn | China | 44 | 58 |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-66670886 |
info@dakenam.com | China | 15928 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 |
fandachem@gmail.com | China | 9348 | 55 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-61398051 +8613650506873 |
sales@chemdad.com | China | 39916 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-29-87569265 +86-18612256290 |
1056@dideu.com | China | 3632 | 58 |
1H,1H,2H,2H-Perfluorooctanesulfonic acid 관련 검색:
헵타데카플루오로-1-옥테인설폰산(헵타데카플루오르-1-옥탄설폰산) 리튬 과플루오로옥탄 황산염 칼륨 1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-헵타데카플루오로-1- 옥탄술폰산 1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-헵타데카플루오로-1- 옥탄술폰 산 화합물 ,함유 2,2'- 이미 노비스 [에탄올] (1 : 1) 헵타데카플루오로 옥탄술폰산 테트라에틸암모늄염 암모늄 1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-십칠불화- 1-옥탄술폰산
Synephrine
Simvastatin
Perfluoroalkylethyl acrylate
(Perfluorohexyl)ethylene
(Perfluorobutyl)ethylene
Perfluoroalkyl Ethylenes
1,1,1,2,2,3,3,4,4,5,5,6,6-Tridecafluoro-8-iodooctane
1H,1H,2H,2H-Perfluorooctyl acrylate
Perfluoro-1-iodohexane
1H,1H,2H-Perfluoro-1-decene
1H,1H,2H,2H-Perfluorohexyl iodide
Perfluoroalkyl alcohol