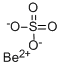베릴륨 황산염 C화학적 특성, 용도, 생산
화학적 성질
Colorless crystals. Soluble in water; insoluble in alcohol.
물리적 성질
Colorless crystalline solid; tetragonal crystal system; hygroscopic; density 2.50 g/cm
3 (tetrahydrate 1.71 g/cm
3); tetrahydrated salt loses water of crystallization on heating; further heating to 550°C causes decomposition; soluble in water, tetrahydrate more soluble in water (30.5g/100g at 30°) than the anhydrous salt; insoluble in alcohol.
용도
No major commercial application of beryllium sulfate is known.
제조 방법
Beryllium sulfate may be prepared by treating an aqueous solution of any beryllium salt with sulfuric acid, followed by evaporation of the solution and crystallization. The hydrated product may be converted to anhydrous salt by heating at 400°C.
정의
ChEBI: A metal sulfate in which the metal is beryllium (in the +2 oxidation state) and the ratio of beryllium to sulfate is 1:1.
일반 설명
Colorless, odorless crystals. Highly toxic by inhalation and ingestion. Sinks and mixes with water.
공기와 물의 반응
Soluble in water.
반응 프로필
BERYLLIUM SULFATE is incompatible with the following: Acids, caustics, chlorinated hydrocarbons, oxidizers, molten lithium .
위험도
A confirmed carcinogen.
건강위험
Any dramatic, unexplained weight loss should be considered as possible first indication of beryllium disease. Other symptoms include anorexia, fatigue, weakness, malaise. Inhalation causes pneumonitis, nasopharyngitis, tracheobronchitis, dyspnea, chronic cough. Contact with eyes causes conjunctival inflammation. Contact with skin causes dermatitis of primary irritant or sensitization type; causes ulcer formation when in contact with cuts.
화재위험
Special Hazards of Combustion Products: Toxic beryllium oxide and sulfuric acid fumes may form in fire.
Safety Profile
Confirmed carcinogen withexperimental tumorigenic data. Acute poison byinhalation, ingestion, intraperitoneal, subcutaneous,intravenous, and intratracheal routes. Mutation data reported. When heated to decomposition itemits very toxic fumes of SOx an
베릴륨 황산염 준비 용품 및 원자재
원자재
준비 용품