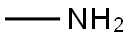메틸아민
|
|
메틸아민 속성
- 녹는점
- -93 °C(lit.)
- 끓는 점
- -6.3 °C(lit.)
- 밀도
- 0.785 g/mL at 25 °C
- 증기 밀도
- 1.08 (20 °C, vs air)
- 증기압
- 27 psi ( 20 °C)
- 굴절률
- n
20/D 1.371
- 인화점
- 61 °F
- 저장 조건
- Store below +30°C.
- 용해도
- highly soluble in water (108g/100g) at 25°C; soluble in alcohol and miscible with ether; HCl salt is soluble in water and absolute alcohol; compound is insoluble in chloroform, acetone, ether, and ethyl acetate
- 산도 계수 (pKa)
- 10.63(at 25℃)
- 물리적 상태
- 가스
- Specific Gravity
- 0.901 (20℃/4℃) (40% Soln.)
- 수소이온지수(pH)
- 14 (H2O, 20°C)
- Odor Threshold
- 0.035ppm
- 폭발한계
- 4.9-20.8%
- 수용성
- 물, 에탄올, 벤젠, 아세톤 및 에테르와 혼합 가능합니다.
- Merck
- 14,6014
- BRN
- 741851
- Henry's Law Constant
- (x 10-5 atm?m3/mol): 1.11 at 25 °C (Christie and Crisp, 1967)
- 노출 한도
- TLV-TWA 10 ppm (~12.3 mg/m3)(ACGIH, MSHA, and OSHA); IDLH 100 ppm (NIOSH).
- Dielectric constant
- 10.0(18℃)
- 안정성
- 안정적인. 가연성이 높습니다. 넓은 폭발 한계에 유의하십시오. 산화제, 산, 알칼리, 알칼리 토금속, 구리 및 그 합금, 아연 및 그 합금과 호환되지 않습니다.
- LogP
- -0.570
- CAS 데이터베이스
- 74-89-5(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | F+,Xn,C,F,T | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 12-20-37/38-41-34-20/22-11-39/23/24/25-36/37/38-23/24/25-40-19 | ||
| 안전지침서 | 7-16-26-36/37-45-29-36/37/39-3/7-3-39-33 | ||
| 유엔번호(UN No.) | UN 3286 3/PG 2 | ||
| WGK 독일 | 2 | ||
| RTECS 번호 | PF6300000 | ||
| F 고인화성물질 | 4.5-31 | ||
| 자연 발화 온도 | 806 °F | ||
| TSCA | Yes | ||
| DOT ClassificationII | 2.1 (Flammable gas) | ||
| 위험 등급 | 3 | ||
| 포장분류 | II | ||
| HS 번호 | 29211100 | ||
| 유해 물질 데이터 | 74-89-5(Hazardous Substances Data) | ||
| 독성 | LD50 orally in rats: 100-200 mg/kg (Kinney); LC50 in rats: 0.448 ml/l (Sarkar, Sastry) | ||
| IDLA | 100 ppm | ||
| 기존화학 물질 | KE-23421 | ||
| 사고대비 물질 필터링 | 7 |
메틸아민 C화학적 특성, 용도, 생산
개요
암모니아의 수소 원자 하나를 메틸기로 치환한 지방족 아민의 하나. 강한 암모니아 냄새가 나는 기체로, 생선이나 고기가 썩을 때 발생한다.용도
가죽을 무두질할 때나 유기 합성의 원료로 쓰인다.화학적 성질
Methylamine is a colorless, fi sh-like smelling gas at room temperature. It is used in a variety of industries, such as the manufacture of dyestuffs, treatment of cellulose, acetate rayon, as a fuel additive, rocket propellant, and in leather tanning processes.물리적 성질
Colorless, flammable gas with a strong ammonia-like odor. An experimentally determined recognition odor threshold concentration of 21 ppbv was reported by Leonardos et al. (1969). Odor threshold concentrations of 4.7 ppmv and 35 ppbv were experimentally determined by Nishida et al. (1979) and Nagata and Takeuchi (1990), respectively.용도
Methylamine is used in dyeing and tanning;in photographic developer, as a fuel additive,and as a rocket propellant. It is also usedin organic synthesis and as a polymerizationinhibitor. It occurs in certain plants, such asMentha aquatica.생산 방법
Several methods are currently used for synthesis of methylamine. Virtually all produce a mixture of primary, secondary, and tertiary amines which can be continuously separated by distillation and extraction. The most commonly used synthesis involves heating ammonium chloride and methyl alcohol (ratio varies from 2:1 to 6:1, depending on desired ratio of amines) to about 300°C in the presence of a catalyst such as zinc chloride. Alternatively, methylamine can be synthesized by heating ammonium chloride and formaldehyde in the presence of H2 and a hydrogenation catalyst such as nickel or platinum. Methylamine is generally marketed as a liquid or a 33% aqueous solution (HSDB 1988).정의
ChEBI: The simplest of the methylamines, consisting of ammonia bearing a single methyl substituent.일반 설명
A colorless gas or a liquid. Pungent fishy odor resembling odor of ammonia. The liquid boils at 20.3°F hence vaporizes rapidly when unconfined. Vapors are heavier than air and may collect in low-lying areas. Easily ignited under most conditions. Under prolonged exposure to intense heat the containers may rupture violently and rocket. Used for making pharmaceuticals, insecticides, paint removers, surfactants, rubber chemicals.공기와 물의 반응
Highly flammable. Very soluble in water; the solutions are strongly basic and therefore corrosive. Liquid fumes in air.반응 프로필
METHYLAMINE neutralizes acids in exothermic reactions to form salts plus water. May be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen is generated in combination with strong reducing agents, such as hydrides.위험도
(Gas and liquid) Dangerous fire risk. Explosive limits in air 5–21%. Strong irritant to tissue. Eye, skin and upper respiratory tract irritant.건강위험
Exposures to methylamine are known to cause adverse health effects among occupational workers. The workers demonstrate symptoms of toxicity that include, but are not limited to, irritation to the eyes, nose, and throat. Studies have indicated that the compound causes injury to the eyes through corneal opacities and edema hemorrhages in the conjunctiva, and injury to the liver. Studies of Guest and Varma indicated no signifi cant deleterious effects on the internal organs or skeletal deformities in experimental mice화재위험
FLAMMABLE. POISONOUS GASES MAY BE PRODUCED IN FIRE. Containers may explode in fire. Flashback along vapor trail may occur. Vapor may explode if ignited in an enclosed area. Toxic nitrogen oxides may be formed. Vapors are heavier than air and may travel considerable distance to a source of ignition and flash back.공업 용도
Methylamine and its hydrochloride salt are widely used in organic synthesis for introducing the methylamino group. In 1976, industrial consumption was 32,000 tons/year. One of its most important uses is in the preparation of amide-type surfactants. It is also used in the preparation of drugs such as adrenaline and synthetic caffeine. It serves as the base for more than twenty commercial products, among those included are photographic developers, insecticides, and antihistamine drugs (Beard and Noe 1981). It is also widely used in tanning and has been used in the separation of aromatics from aliphatic hydrocarbons (Sittig 1981).Safety Profile
Poison by subcutaneous route. Moderately toxic by inhalation. A severe skin irritant. Mutation data reported. A strong base. Flammable gas at ordinary temperature and pressure. Very dangerous fire hazard when exposed to heat, flame, or sparks. Explosive when exposed to heat or flame. To fight fire, stop flow of gas. Forms an explosive mixture with nitromethane. When heated to decomposition it emits toxic fumes of NOx. See also AMINES.잠재적 노출
Methylamine is used in organic synthesis; a starting material for N-oleyltaurine, a surfactant; and p-N-methylaminophenol sulfate, a photographic developer. It has possible uses in solvent extraction systems in separation of aromatics from aliphatic hydrocarbons. It is also used in the synthesis of many different pharmaceuticals; pesticides and rubber chemicals.Carcinogenicity
Methylamine was positive in the mouse lymphoma assay and negative in the Ames assay.환경귀착
Photolytic. The rate constant for the reaction of methylamine and OH radicals in the atmosphere at 300 K is 1.3 x 10-13 cm3/molecule?sec (Hendry and Kenley, 1979).Low et al. (1991) reported that the photooxidation of aqueous primary amine solutions by UV light in the presence of titanium dioxide resulted in the formation of ammonium and nitrate ions.
Chemical/Physical. In an aqueous solution, chloramine reacted with methylamine to form Nchloromethylamine (Isaac and Morris, 1983).
Reacts with acids forming water-soluble salts.
저장
Methylamine is stored in a cool, well-ventilated noncombustible area separatedfrom possible sources of ignition andoxidizing substances and mercury. Itssolutions are stored in a flammable liquidstorage room or cabinet. The gas is shippedin steel cylinders or tank cars; the liquid isshipped in steel drums or tank cars.운송 방법
UN1061 Methylamine, anhydrous, Hazard Class: 2.1; Labels: 2.1-Flammable gas. UN1235 Methylamine, aqueous solution, Hazard Class: 3; Labels: 3-Flammable liquid, 8-Corrosive material. Cylinders must be transported in a secure upright position, in a well-ventilated truck. Protect cylinder and labels from physical damage. The owner of the compressed gas cylinder is the only entity allowed by federal law (49CFR) to transport and refill them. It is a violation of transportation regulations to refill compressed gas cylinders without the express written permission of the owner.Purification Methods
Dry the amine with sodium or BaO. It is commercially available in metal cylinders. [Beilstein 4 IV 118.]비 호환성
A medium-strong base. Reacts violently with strong acids; mercury, strong oxidizers; nitromethane. Corrosive to copper, zinc alloys; aluminum, and galvanized surfaces.폐기물 처리
Return refillable compressed gas cylinders to supplier. Controlled incineration (incinerator equipped with a scrubber or thermal unit to reduce nitrogen oxides emissions).메틸아민 준비 용품 및 원자재
원자재
준비 용품
N-Methyl-4-pyridinamine
3-Hydrazinyl-N-methylpropanamide
N-Cyano-N'-methyl-ethanimidamide
sodium oleyl sarcosinate
additive AC1210
fluorescent whitening agent AT
didodecyl dimethyl ammoniumm
Methoxyphenamine
1-Methyl-3-phenyl-2-thiourea
BIFEMELANE
N,N-DIMETHYL-4-[(METHYLAMINO)METHYL]ANILINE
Noncyanide zinc plating additive DE
3,4,5-TRIMETHACARB
METHYL-(2-TRIFLUOROMETHYL-BENZYL)-AMINE
CHLOROMETHYLACETAMIDE
4-Chloro-N-methylaniline
(4-FLUORO-BENZYL)-METHYL-AMINE
N-Methylpentylamine
(3-FLUOROBENZYL)METHYLAMINE
C.I. 용제 적 111
2-CHLORO-N-METHYLACETOACETAMIDE
Methylchloroisothiazolinone/methylisothiazolinone mixture (MCIT/MIT)
다이메틸우레아
졸피뎀
로테논
2-AMINO-4-METHYLAMINO-6-ETHOXY-1,3,5-TRIAZINE
(1H-IMIDAZOL-2-YLMETHYL)-METHYL-AMINE
3-METHOXY-N-METHYLBENZYLAMINE 97
Bonnecor
2-Hydrazinyl-N-methylacetamide
2-Methoxy-N-methylbenzylamine hydrochloride ,97%
(2-FLUOROBENZYL)METHYLAMINE
N-(4-Methoxybenzyl)-N-methylamine
POLY DIMETHYL DIALLYL AMMONIUM CHLORIDE
Rilmazafone
3-CHLORO-N-METHYLBENZYLAMINE
N-METHYLURETHANE
메티마졸
METHYL-PYRIDIN-2-YLMETHYL-AMINE
1-Methyl-3-pyrrolidinol