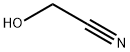
포름알데히드 시아노히드린 C화학적 특성, 용도, 생산
화학적 성질
light yellow liquid (typically available as a concentrated. Glycolonitrile [107-16-4], hydroxyacetonitrile, formaldehyde cyanohydrin, HOCH2CN, Mr
57.05, bp 183℃ (slight decomp.), d
154 1.104, n
20D1.4117. Anhydrous glycolonitrile is a colorless
liquid which is miscible with water, ethanol and
diethyl ether.
용도
Solvent and organic intermediate.
생산 방법
Glycolonitrile is the result of reaction between formaldehyde
and aqueous sodium cyanide in the presence of mineral acid.
일반 설명
Odorless colorless oil with a sweetish taste. Used in the manufacture of intermediates in pharmaceutical production, as a component of synthetic resins, as a chemical intermediate for organic compounds, and as a solvent.
공기와 물의 반응
Water soluble.
반응 프로필
Glycolonitrile may undergo spontaneous and violent decomposition. Traces of alkali (base) promote violent polymerization [Lewis].
위험도
Toxic by ingestion, inhalation, and skin
absorption.
건강위험
Extremely toxic, exposure by any route should be avoided; may have fatal consequences; death from asphyxiation may occur similar to that resulting from hydrogen cyanide.
화재위험
Moderate explosion hazard when exposed to heat or by spontaneous chemical reaction in the presence of alkalies if uninhibited. When heated to decomposition, Glycolonitrile emits highly toxic fumes of cyanide and nitrogen oxides. Unstable, may explode on standing. Hazardous polymerization may occur. avoid the presence of alkalis, and exposure to heat.
잠재적 노출
Formaldehyde cyanohydrin is used in
the manufacture of intermediates in pharmaceutical produc tion and as a component of synthetic resins as a chemical
intermediate for organic compounds, and as a solvent.
운송 방법
UN3276 Nitriles, liquid, toxic, n.o.s., Hazard
Class: 6.1; Labels: 6.1-Poisonous materials, Technical
Name Required, Potential Inhalation Hazard (Special
Provision 5). UN2810 Toxic liquids, organic, n.o.s., Hazard
Class: 6.1; Labels: 6.1-Poisonous materials, Technical
Name Required.
비 호환성
Incompatible with oxidizers (chlorates,
nitrates, peroxides, permanganates, perchlorates, chlorine,
bromine, fluorine, etc.); contact may cause fires or explo sions. Keep away from alkaline materials, strong bases,
strong acids, oxoacids, epoxides, and exposure to heat.
Unless stabilized with a weak acid solution, traces of alka lis may cause violent polymerization.
포름알데히드 시아노히드린 준비 용품 및 원자재
원자재
준비 용품