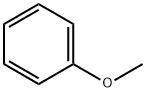
아니졸 C화학적 특성, 용도, 생산
개요
Anisole is a colorless liquid with a spicy-sweet smell. Anisole can be produced by all the aforementioned methods. It is an important intermediate in the synthesis of organic compounds, for example fragrances and pharmaceuticals. The compound is also used as a solvent and as a heat transfer medium.
화학적 성질
Anisole is a colorless to yellowish liquid with a characteristic pleasant, anise-like, agreeable, aromatic, spicy-sweet odor. It is used in perfumery.
출처
Anisole is a natural product found in apple juice and in the oil of Artemisia dracunculus var. turkestanica; also reported found in butter, Camembert cheese, roasted beef, olive (Olea europae), Malay apple, Jerusalem artichoke (Helianthus tuberosus), Bourbon vanilla, truffles, crab and sopadilla fruit (Achras sapota L.).
용도
Anisole is widely used as a solvent for the synthesis of various organic compounds, anethole, nonylphenol isomer 4-(3',6'-dimethyl-3-heptyl)phenol, perfumes, insect pheromones and pharmaceuticals. It finds application in the preparation of inorganic complexes and materials such as tin-core/tin oxide nanoparticles.
주요 응용
Anisole is an organic compound with the chemical formula C7H8O with a pleasant anise-like aroma, used in organic synthesis and also as a solvent, fragrance and insect repellent. For organic synthesis, it is also used as solvent, fragrance and insect repellent.
제조 방법
Anisole is synthesized by reacting phenol and dimethyl sulfate in the presence of aqueous NaOH; by passing methyl chloride into a suspension of sodium phenolate in liquid ammonia.
정의
ChEBI: Anisole is a monomethoxybenzene that is benzene substituted by a methoxy group. It has a role as a plant metabolite.
일반 설명
A clear straw-colored liquid with an aromatic odor. Insoluble in water and the same density as water. Vapors heavier than air. Flash point 125°F. Boiling point 307°F. Moderately toxic by ingestion. A skin irritant. Used to make perfumes, flavorings and as a solvent.
공기와 물의 반응
Flammable. Ethers tend to form unstable peroxides when exposed to oxygen. Ethyl, isobutyl, ethyl tert-butyl, and ethyl tert-pentyl ether are particularly hazardous in this respect. Ether peroxides can sometimes be observed as clear crystals deposited on containers or along the surface of the liquid. Insoluble in water
반응 프로필
Ethers, such as Anisole can act as bases. They form salts with strong acids and addition complexes with Lewis acids. The complex between diethyl ether and boron trifluoride is an example. Ethers may react violently with strong oxidizing agents. In other reactions, which typically involve the breaking of the carbon-oxygen bond, ethers are relatively inert.
건강위험
Inhalation or contact with material may irritate or burn skin and eyes. Fire may produce irritating, corrosive and/or toxic gases. Vapors may cause dizziness or suffocation. Runoff from fire control or dilution water may cause pollution.
Safety Profile
Moderately toxic by
ingestion and inhalation. A skin irritant. A
flammable liquid. To fight fire, use foam,
CO2, dry chemical. When heated to
decomposition it emits acrid fumes.
잠재적 노출
Anisole is used as a solvent; a flavoring,
vermicide, making perfumes; and in organic synthesis.
Purification Methods
Shake anisole with half its volume of 2M NaOH, and the emulsion is allowed to separate. Repeat three times, then wash twice with water, dry over CaCl2, filter, dry over sodium wire and finally distil it from fresh sodium under N2 using a Dean-Stark trap (samples in the trap being rejected until free from turbidity) [Caldin et al. J Chem Soc, Faraday Trans 1 72 1856 1976]. Alternatively dry it with CaSO4 or CaCl2, or by refluxing with sodium or BaO with crystalline FeSO4 or by passage through an alumina column. Traces of phenols are removed by prior shaking with 2M NaOH, followed by washing with water. It has been be purified by zone refining. [Beilstein 6 IV 548.]
비 호환성
Incompatible with oxidizers (chlorates,
nitrates, peroxides, permanganates, perchlorates, chlorine,
bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong
bases, strong acids, oxoacids, epoxides.
폐기물 처리
Dissolve or mix the material
with a combustible solvent and burn in a chemical incinerator
equipped with an afterburner and scrubber. All federal,
state, and local environmental regulations must be
observed.
아니졸 준비 용품 및 원자재
원자재
준비 용품
cleaner LS
4,5-이미다졸디카르복시산
6-METHOXY-1-NAPHTHOIC ACID
4-chloro-4'-methoxybenzophenone
6-Hydroxy-1-naphthoic acid
PYRIDIN-2-YLMETHYL-HYDRAZINE
Fast Scarlet Base LG
3-(4-METHOXYBENZOYL)PROPIONIC ACID
Diaveridine
1-Methyl-1H-imidazole-4-carboxylic acid
메톡시클로르
4(5)-(히드록시메틸)이미다졸
4,5-DICARBOXY-1-METHYL-1H-IMIDAZOLE
Cefcapene pivoxil
PYRIDIN-3-YLMETHYL-HYDRAZINE
3,4-다이하이드록시-알파-메틸페닐알라닌
4,5-Dicyanoimidazole
Verapamil
4-이미다졸카르복시산
trisodium [5-acetamido-4-hydroxy-3-[[2-hydroxy-5-[[2-(sulphooxy)ethyl]sulphonyl]phenyl]azo]naphthalene-2,7-disulphonato(5-)]cuprate(3-)
1-CHLOROMETHYL-2,3,4-TRIMETHOXYBENZENE
1,5-디아자비시클로[4.3.0]논-5-엔
4-메톡시 벤질아세테이트
2-브로모아니솔
PYRIDIN-4-YLMETHYL-HYDRAZINE
Ceftibuten
2,4,6-트리브로모아니졸
Cefdinir
Lawesson 시약
Anoxomer
식용 적색 40호
Fenofibrate
4-Fluoro-4'-methoxybenzophenone
4-Chloro-4'-hydroxybenzophenone
C.I. 디스페르제 BLUE 73
1-METHYL-1H-IMIDAZOLE-5-CARBOXYLIC ACID
Bunazosin hydrochloride
Alacepril
CHLOROTRIANISENE
1-METHYL-1H-IMIDAZOLE-4,5-DICARBONITRILE