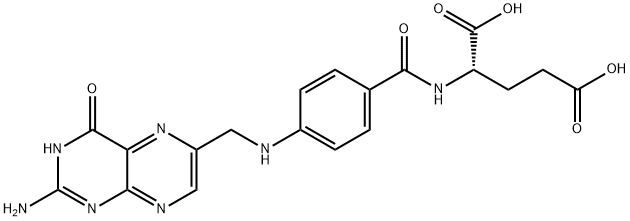
엽산
|
|
엽산 속성
- 녹는점
- 250 °C
- 알파
- 20 º (c=1, 0.1N NaOH)
- 끓는 점
- 552.35°C (rough estimate)
- 밀도
- 1.4704 (rough estimate)
- 굴절률
- 1.6800 (estimate)
- 저장 조건
- 2-8°C
- 용해도
- 끓는 물: 가용성1%
- 물리적 상태
- 결정성 분말
- 산도 계수 (pKa)
- pKa 2.5 (Uncertain)
- 색상
- 노란색에서 주황색으로
- 냄새
- 냄새 없는
- pH 범위
- 4
- 수용성
- 1.6mg/L(25℃)
- Merck
- 14,4221
- BRN
- 100781
- BCS Class
- 3
- 안정성
- 안정적인. 중금속 이온, 강한 산화제, 강한 환원제와 호환되지 않습니다. 용액은 가볍고 열에 민감할 수 있습니다.
- InChIKey
- OVBPIULPVIDEAO-LBPRGKRZSA-N
- LogP
- -0.990 (est)
- CAS 데이터베이스
- 59-30-3(CAS DataBase Reference)
- NIST
- Folic acid(59-30-3)
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험 카페고리 넘버 | 33-62-68 | ||
|---|---|---|---|
| 안전지침서 | 24/25 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | LP5425000 | ||
| F 고인화성물질 | 8 | ||
| TSCA | Yes | ||
| HS 번호 | 29362900 | ||
| 유해 물질 데이터 | 59-30-3(Hazardous Substances Data) | ||
| 독성 | A water soluble vitamin required in the diet of mammals. Sulfonamide drugs are selectively toxic to bacteria because they inhibit the incorporation of p-aminobenzoic acid into folic acid, a biosynthetic process in bacteria. Folic acid deficiency adversely affects prenatal development in humans. Dietary supplementation with folic acid dramatically reduces the incidence of neural tube defects in humans. Folic acid deficiency may also contribute to the causes of megaloblastic macrocytic anemia and a consequence of this is that this disease can be induced, as a side effect, when methotrexate, a folic acid antagonist, is used in cancer chemotherapy | ||
| 기존화학 물질 | KE-01305 |
| 그림문자(GHS): |

|
||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 신호 어: | Warning | ||||||||||||||||||||||||||||
| 유해·위험 문구: |
|
||||||||||||||||||||||||||||
| 예방조치문구: |
|
엽산 C화학적 특성, 용도, 생산
존재
엽산은 말린 콩, 완두, 렌즈 콩, 오렌지, 간, 아스파라거스, 사탕무, 브로콜리, 브뤼셀 콩나물, 시금치와 같은 식품에서 쉽게 발견 할 수있는 B 비타민 (비타민 B9)의 한 종류입니다. 새로운 건강한 세포를 만드는 능력으로, 엽산은 임산부에게 중요합니다. 임신 전이나 임신 중 엽산 결핍은 아기의 척추 또는 뇌의 주요 출생 결함을 예방할 수 있습니다.개요
물에서 불용해성 공기 & 물 반응. 수성 해결책에는 4.0-4.8의 pHs가 있습니다.엽산의 반응성 단면도 산성 해결책은 열에 과민합니다, 그러나 중립성으로, 안정성은 진보적으로 증가합니다. 해결책은 자외선에 의해 활동되지 않고 알칼리성 해결책은 산화에 과민합니다. 엽산은 또한 빛에 의해 활동되지 않ㅂ니다. 엽산은 산화제, 감소시키는 대리인 및 중금속 이온에 상반됩니다. 엽산의 화재의 원인이 되는 것 인화점 데이터는 유효하지 않습니다; 그러나, 엽산은 아마 가연성 입니다
용도
엽산 (비타민 B9)은 엽산이라고 불리는 비타민 B를 인간이 만들었습니다. 우리 몸이 적혈구를 생성하는 것은 매우 중요합니다. 엽산은 강화 시리얼에 존재합니다. 임산부 또는 임신 준비가되어있는 사람들에게 널리 사용됩니다. 엽산 (비타민 B9)은 유산이나 아기의 자연적인 굴절 또는 조기 진통의 위험을 크게 줄입니다. 엽산 보충제는 모든 의사의 조언에 의한 것입니다.모든 사람의 건강을 위해 엽산 결핍으로 빈혈이 유발됩니다. 엽산은 다른 약물과 함께 사용하거나 혼자서이 문제를 치료할 수 있습니다. 예 : 푸마르산 철 및 엽산 정
신장 질환, 빈혈로 고통 받고 있거나 대개 많이 마시면 의사의 전문 지식을 따라 엽산을 섭취하십시오.
제품 소개
엽산은 말린 콩, 완두, 렌즈 콩, 오렌지, 간, 아스파라거스, 사탕무, 브로콜리, 브뤼셀 콩나물, 시금치와 같은 식품에서 쉽게 발견 할 수있는 B 비타민 (비타민 B9)의 한 종류입니다. 새로운 건강한 세포를 만드는 능력으로, 엽산은 임산부에게 중요합니다. 임신 전이나 임신 중 엽산 결핍은 아기의 척추 또는 뇌의 주요 출생 결함을 예방할 수 있습니다.엽산 (비타민 B9) 원료 인 API는 새로운 세포를 생산하고 유지하는 데 도움이되며 암으로 이어질 수있는 DNA의 변화를 막아줍니다. 제약 분야에서 엽산은 주로 엽산 결핍으로 인한 엽산 (비타민 B9) 결핍 및 특정 유형의 빈혈 (적혈구 부족)을 치료하는 데 사용됩니다. 때로는 엽산 (비타민 B9)이 다른 약물과 함께 악성 빈혈을 치료합니다. 우리가 더 잘 알아야 할 것은, 엽산 (비타민 B9)이 비타민 B12 결핍증에 효과적이지 않으며 척수 손상을 예방하지 않는다는 것입니다.
순도시험
(1) 유리아민 : 파라아미노벤조일글루탐산표준품을 감압데시케이타(실리카겔)에서 4시간 건조한 다음 약 0.05g을 정밀히 달아 에탄올(2→5)을 가하여 녹여 100mL로 하고 그 중 5mL를 취하여 물을 가해 1,000mL로 한다. 그 중 4mL 및 물 5mL를 취하여 각각의 정량법의 B액으로부터 시험용액을 만드는 조작과 같이 조작하여 만든 액을 각각 S''액 및 D액으로 한다. D액을 대조액으로 하여 S''액에 대해 파장 550nm에서의 흡광도를 측정하여 얻은 수를 AS''로 한다. AS''와 정량법에서 얻은 Ac로 부터 다음 계산식에 따라 유리아민의 양을 구할 때, 그 값은 1% 이하이어야 한다.
(2) 납 : 이 품목 5.0g을 취하여 원자흡광광도법 또는 유도결합플라즈마발광광도법에 따라 시험할 때, 그 양은 2.0ppm 이하이어야 한다.
확인시험
이 품목 1.5mg에 수산화나트륨용액(1→250)을 가하여 녹인 다음 100mL로 한 액은 파장 255~257nm, 281~285nm 및 361~369nm에 극대흡수부가 있다.
정량법
이 품목 0.1g을 정밀히 달아 0.1N 수산화나트륨용액을 가하여 잘 흔들어 섞어 녹이고 200mL로 한다. 그 중 25mL를 취하여 묽은염산 20mL 및 물을 가하여 100mL로 하고 이를 A액으로 한다. A액 60mL를 취하여 아연말 0.5g을 가하고 때때로 흔들어 섞으면서 20분간 방치한다. 이어서 건조여과지로 여과하여 처음 여액 약 10mL를 버리고 나머지 여액 10mL를 취하여 물을 넣어 100mL로 한 액을 B액으로 한다. B액 5mL를 취하여 묽은염산 1mL 및 아질산나트륨용액(1→1,000) 1mL를 가하고 잘 흔들어 섞은 다음 2분간 방치하고 이어서 설파민산암모늄용액(1→200) 1mL를 가하고 잘 흔들어 섞은 다음 2분간 방치한다. 이에 N-(1-나프틸)-N-(디에틸)-에틸렌디아민수산염용액(1→1,000) 1mL를 가하고 흔들어 섞은 다음 10분간 방치하고 물을 가하여 20mL로 하고 이를 시험용액으로 한다. A액 5mL를 취하여 이하 B액으로부터 시험용액을 만드는 조작과 같이 만든 액을 C액으로 한다. 엽산표준품 약 0.1g을 정밀히 달아 이하 검체로부터 시험용액을 만드는 조작과 같이 조작하여 만든 액을 표준용액으로 한다. 따로 물 5mL를 취하여 이하 B액으로부터 시험용액을 만드는 조작과 같이 조작하여 대조액을 만들고 이를 대조하여 시험용액, C액 및 표준용액에 대해 파장 550nm에 있어서의 흡광도를 측정하여 얻은 수치를 각각 A, AC 및 AS로 하여 다음 계산식에 따라 엽산의 함량을 구한다. 다만, 엽산표준품 및 검체의 수분의 측정은 수분에 정하여진 방법에 의한다.
개요
Folic acid is an essential B vitamin. It is converted to folate in vivo, which is a necessary cofactor for a variety of biological processes, including nucleotide synthesis and, thus, DNA synthesis and repair, among others. A deficiency in dietary folic acid can lead to a range of developmental and cognitive disorders, most prominently neural tube defects and congenital heart defects.화학적 성질
orange to yellow crystalline powder물리적 성질
The folates include a large number of chemically related species, each differing with respect to the various substituents possible at three sites on the pteroylglutamic acid basic structure. More than 170 different folates are theoretically possible. Not all of these occur in nature; but it has been estimated that as many as 100 different forms are found in animals. The folates from most natural sources usually have a single carbon unit at N-5 and/or N-10; these forms participate in the metabolism of the single-carbon pool.Folic acid (pteroylmonoglutamic acid) is an orange-yellow crystalline substance that is soluble in water but insoluble in ethanol or less polar organic solvents. It is unstable to light, to acidic or alkaline conditions, to reducing agents, and, except in dry form, to heat. It is reduced in vivo enzymatically (or in vitro with a reductant such as dithionite) first to 7,8-dihydrofolic acid (FH2) and then to FH4; both of these compounds are unstable in aerobic environments and must be protected by the pres ence of an antioxidant (e.g., ascorbic acid, 2-mer
Two derivatives of folic acid, each having an amino group in the place of the hydroxyl at C-4, are folate antagonists of biomedical use: aminopterin (4-aminofolic acid) and methotrexate (4-amino-N10-methylfolic acid). Aminopterin is used as a rodenticide; methotrexate is an antineoplastic agent.
출처
Synthetic용도
folic acid is generally used as an emollient. In vitro and in vivo skin studies now indicate its capacity to aid in DnA synthesis and repair, promote cellular turnover, reduce wrinkles, and promote skin firmness. There is some indication that folic acid may also protect DnA from uV-induced damage. Folic acid is a member of the vitamin B complex and is naturally occurring in leafy greens.정의
ChEBI: An N-acyl-amino acid that is a form of the water-soluble vitamin B9. Its biologically active forms (tetrahydrofolate and others) are essential for nucleotide biosynthesis and homocysteine remethylation.일반 설명
Odorless orange-yellow needles or platelets. Darkens and chars from approximately 482°F.공기와 물의 반응
Insoluble in water. Aqueous solutions have pHs of 4.0-4.8.반응 프로필
Acid solutions of Folic acid are sensitive to heat, but towards neutrality, stability progressively increases. Solutions are inactivated by ultraviolet light and alkaline solutions are sensitive to oxidation. Folic acid is also inactivated by light. Folic acid is incompatible with oxidizing agents, reducing agents and heavy metal ions.화재위험
Flash point data for Folic acid are not available; however, Folic acid is probably combustible.Safety Profile
Poison by intraperitoneal and intravenous routes. Experimental teratogenic effects. Mutation data reported. When heated to decomposition it emits toxic fumes of NOx.Purification Methods
If paper chromatography indicates impurities, then recrystallise it from hot H2O or from dilute acid [Walker et al. J Am Chem Soc 70 19 1948]. Impurities may be removed by repeated extraction with n-BuOH of a neutral aqueous solution of folic acid (by suspending in H2O and adding N NaOH dropwise till the solid dissolves, then adjusting the pH to ~7.0-7.5) followed by precipitation with acid, filtration, or better collected by centrifugation and recrystallised form hot H2O. [Blakley Biochem J 65 331 1975, Kalifa et al. Helv Chim Acta 6 1 2739 1978.] Chromatography on cellulose followed by filtration through charcoal has also been used to obtain pure acid. [Sakami & Knowles Science 129 274 1959.] UV: max 247 and 296nm ( 12,800 and 18,700) in H2O pH 1.0; 282 and 346nm ( 27.600 and 7,200) in H2O pH 7.0; 256, 284 and 366nm ( 24600, 24,500 and 86,00) in H2O pH 13 [Rabinowitz in The Enzymes (Boyer et al. Eds), 2 185 1960]. [Beilstein 26 III/IV 3944.]엽산 준비 용품 및 원자재
원자재
준비 용품
엽산 공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hengshui Haoye Chemical Co.,Ltd. | +86-2102300 +86-18632882519 |
hy@chemcoms.com | China | 256 | 58 |
| TAIZHOU YUXIN BIOTECHNOLOGY CO,.LTD | +86-576-88902229;+86-0576-88902229 +8613968687450 |
yuxin@yuxchem.com | China | 122 | 58 |
| XI AN SGONEK BIOLOGICAL TECHNOLOGY CO., LTD | 15202951039 |
jason@sgonekbio.com | China | 268 | 58 |
| Wuhan Boyuan Import & Export Co., LTD | +8615175982296 |
Mike@whby-chem.com | China | 974 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12453 | 58 |
| Hebei Dongdu Import and Export Co. LTD | +86-15333296769 +86-15333296769 |
manager@cndongdu.com | China | 71 | 58 |
| shandong perfect biotechnology co.ltd | +86-53169958659; +8618596095638 |
sales@sdperfect.com | China | 294 | 58 |
| Across Biotech Jinan Co LTD | +8613031735486 |
frank@acrossbiotech.com | China | 105 | 58 |
| Nantong Guangyuan Chemicl Co,Ltd | +undefined17712220823 |
admin@guyunchem.com | China | 616 | 58 |
| Hebei Kangcang new material Technology Co., LTD | +8619133911216 |
Jany1001@kangcang.com.cn | China | 338 | 58 |