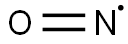산화 질소
|
|
산화 질소 속성
- 녹는점
- −163.6 °C(lit.)
- 끓는 점
- −151.7 °C(lit.)
- 밀도
- d-150.2 (liq) 1.27; Relative d (gas) 1.036 (air = 1); Absolute d (gas) 1.227 (air = 1)
- 증기 밀도
- 1.05 (vs air)
- 굴절률
- nD25 1.0002697
- 용해도
- 20°C, 101kPa의 압력에서 1부피는 약 21부피의 물에 용해됩니다.
- 물리적 상태
- 무색 가스
- 색상
- 무색의
- 수용성
- 물에 거의 녹지 않음 [HAW93]
- Merck
- 13,6611
- 노출 한도
- TLV-TWA 25 ppm (~30 mg/m3) (ACGIH, MSHA, and OSHA).
- 안정성
- 공기 중의 산소와 자발적으로 반응하여 갈색 이산화질소를 생성합니다. 암모니아 및 많은 유기 물질과 격렬하게 또는 폭발적으로 반응합니다.
- InChIKey
- MWUXSHHQAYIFBG-UHFFFAOYSA-N
- CAS 데이터베이스
- 10102-43-9(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | O,T | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 8-23-34-44 | ||
| 안전지침서 | 17-23-36/37/39-45 | ||
| 유엔번호(UN No.) | UN 1660 2.3 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | QX0525000 | ||
| DOT ClassificationII | 2.3, Hazard Zone A (Gas poisonous by inhalation) | ||
| 위험 등급 | 2.3 | ||
| 유해 물질 데이터 | 10102-43-9(Hazardous Substances Data) | ||
| 독성 | LCLo inhalation in dog: 5000ppm/25M | ||
| IDLA | 100 ppm | ||
| 기존화학 물질 | KE-25996 | ||
| 사고대비 물질 필터링 | 57 |
산화 질소 C화학적 특성, 용도, 생산
개요
near room temperature (its liquid density at 20°C is 1.45 g/cm3). Nitrogen monoxide (NO) is commonly called nitric oxide,Nitric oxide is colorless and has a sharp sweet odor;Nitric oxide is nonfl ammable, toxic gases.Nitric oxide is a free radical that quickly reacts in air to produce nitrogen dioxide.It is also an important biological messenger and transmitter.화학적 성질
Nitric oxide,NO, also known as nitrogen oxide and nitrogen monoxide, is a colorless gas that will react with oxygen at room temperature to form nitrogen dioxide, N202, a reddish-brown gas.It is soluble in water and alcohol and is used primarily to form other compounds.물리적 성질
Colorless gas; paramagnetic; density 1.3402 g/L; slightly heavier than air, air density 1.04 (air=1); liquefies at -151.8°C to a blue liquid; the refractive index of the liquid 1.330 at -90°C; the density of the liquid 1.269 g/mL at -150.2°C; solidifies at -163.6°C to a bluish-white snow-like solid; critical temperature -94°C; critical pressure 65 atm; slightly soluble in water, 4.6 mL gas dissolves in 100 mL water at 20°C while 7.34 mL and 2.37 mL dissolve in the same volume of water at 0 and 60°C, respectively; more soluble in alcohol than water; soluble in carbon disulfide, and in ferrous sulfate solution (reacts).역사
Nitric oxide was prepared in 1772 by Joseph Priestley (1733–1804) and described in his volumes Experiments and Observations of Different Kinds of Air published between 1774 and 1786. Priestley called nitric oxide nitrous air, nitrogen dioxide nitrous acid vapor, and nitrous oxide phlogisticated nitrous air, but also referred to the latter as diminished nitrous air. He observed the change of clear nitric oxide to red nitrogen dioxide. Priestley prepared nitric oxide by reacting nitric acid with a metal such as copper: 3Cu(s) + 8HNO3(aq) → 2NO(g) + 3Cu(NO3)2(aq) + 4H2O(l).용도
Nitric oxide is used as an intermediate in themanufacture of nitric acid, in the preparationof metal nitrosyls, in bleaching of rayon,and in incandescent lamps. It is produced byheating air at high temperatures.생산 방법
Nitric oxide is commercially produced by the catalytic oxidation of ammonia using aplatinum catalyst: 4NH3(g) + 5O2(g) → 4NO(g) + 6H2O(g).정의
ChEBI: Nitric Oxide is a nitrogen oxide which is a free radical, each molecule of which consists of one nitrogen and one oxygen atom.일반 설명
A colorless gas. Noncombustible but accelerates the burning of combustible material. Vapors heavier than air. Very toxic by inhalation and skin absorption. Heating the containers may cause them to rupture violently and rocket.Nitric oxide was discovered by Van Helmont in 1620. It occurs in the exhaust gases from automobiles along with other oxides of nitrogen, at trace concentrations. It also is found in minute quantities in the upper atmosphere, resulting from the oxidation of nitrogen in the presence of ionizing radiation or by electric discharge. Nitric oxide is the most stable oxide of nitrogen. It is used as an intermediate or as a starting reactant in the production of many nitrogen compounds, including nitrogen dioxide, nitric acid and nitrosyl chloride.
공기와 물의 반응
Combines very rapidly with oxygen in the air to form nitrogen dioxide. Nitrogen dioxide reacts with water to form nitric acid and NITRIC OXIDE, reacts with alkalis to form nitrates and nitrites [Merck 11th ed. 1989].반응 프로필
NITRIC OXIDE can serve as both an oxidizing agent and as a reducing agent. Sustains the combustion of powdered aluminum [Mellor 5:209-212. 1946-47]. Enflames or explodes when mixed with vapors of carbon disulfide [Mellor 8, Supp. 2:232. 1967]. Reacts vigorously with sodium monoxide above 100°C [Mellor 2, Supp. 2:629. 1961]. Reacts on contact with oxygen at room temperature to form brown gaseous nitrogen dioxide. Reacts with alkalis to form nitrates and nitrites [Merck 11th ed. 1989]. The liquid is very sensitive to detonation in the presence of water.위험도
Supports combustion. Toxic by inhalation, strong irritant to skin and mucous membranes. Hypoxia/cyanosis, nitrosyl-hemoglobin formation, and upper respiratory tract irritant.건강위험
Can cause death or permanent injury after a very short exposure to small quantities. Irritant of eyes, nose, throat; can cause unconsciousness. NITRIC OXIDE forms acids in the respiratory system which are irritating and cause congestion in the lungs. Concentrations of 60-150 ppm cause immediate irritation of the nose and throat with coughing and burning in the throat and chest. 6-24 hours after exposure, labored breathing and unconsciousness may result. Concentrations of 100-150 ppm are dangerous for short exposure of 30-60 minutes. Concentrations of 200-700 ppm may be fatal after very short exposure.화재위험
Noncombustible gas; burns with fuels, hydrocarbons, or when heated with hydrogen. Nitric oxide reacts violently with carbon disulfide vapors, producing green luminous flame; with fluorine, it produces a pale yel low flame. It explodes when mixed with ozone, chlorine monoxide, or a nitrogen tri halide. Reactions with many pyrophoric met als produce incandescence. Reaction with amorphous boron produces brilliant flashes.Materials Uses
Nitric oxide is noncorrosive, and most common structural materials may be used. However, in the presence of moisture and oxygen, corrosive conditions will develop as a result of the formation of nitric and nitrous acids. Prior to use, systems to contain nitric oxide must first be purged with an inert gas. Where air contamination cannot be eliminated, stainless steel should be used.잠재적 노출
Nitric oxide is used in the manufacture of nitric acid; it is also used in the bleaching of rayon; it is a raw material for nitrosyl halide preparation.환경귀착
Nitric oxide is converted spontaneously in the air to nitrogen dioxide; hence, some of the latter gas is present whenever nitric oxide is found in air (at concentrations below 50 ppm). Nitric oxide is a contributor to photochemical air pollution.저장
Nitric oxide should only be used in well-ventilated areas. Valve protection caps and valve outlet threaded plugs must remain in place unless the container is secured and the valve outlet piped to the point of use. Do not drag, slide, or roll cylinders. Use a suitable hand truck to move cylinders. Use a pressure reducing regulator when connecting a cylinder to lower pressure (1000 psig or 6900 kPa) piping systems. Do not heat a cylinder of nitric oxide by any means to increase the discharge rate from the cylinder. Use a check valve or trap in the discharge line to prevent hazardous reverse flow into the cylinder.운송 방법
UN1660/124 Nitric oxide, compressed, Hazard Class: 2.3; Labels: 2.3-Poisonous gas, 5.1-Oxidizer, 8-Corrosive material, Inhalation Hazard Zone A. Cylinders must be transported in a secure upright position, in a wellventilated truck. Protect cylinder and labels from physical damage. The owner of the compressed gas cylinder is the only entity allowed by federal law (49CFR) to transport and refill them. It is a violation of transportation regulations to refill compressed gas cylinders without the express written permission of the owner.Purification Methods
Bubble the gas through 10M NaOH which removes NO2. It can also be freed from NO2 by passage through a column of Ascarite followed by a column of silica gel held at -197oK. The gas is dried with solid NaOH pellets or by passing through silica gel cooled at -78o, followed by fractional distillation from a liquid N2 trap. This purification does not eliminate nitrous oxide. Other gas scrubbers sometimes used include one containing conc H2SO4 and another containing mercury. It is freed from traces of N2 by the freeze and thaw method. [Blanchard Inorg Synth II 126 1946, Schenk in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I pp 485-487 1963.] TOXIC.비 호환성
A strong oxidizer but may also act as a reducing agent. Explosive reaction with nitrogen trichloride, ozone, carbon disulfide; pentacarbonyl iron; chlorine monoxide. Incompatible with halogens, combustibles, metals, oil, alcohols, chlorinated hydrocarbons (e.g., trichloroethylene), reducing agents (such as NH3), oxygen, fluorine, metals. Reacts with water to form nitric acid. Rapidly converted in air to nitrogen dioxide. Combines very rapidly with oxygen in the air to form nitrogen dioxide. Nitrogen dioxide reacts with water to form nitric acid and nitric oxide, reacts with alkalis to form nitrates and nitrites.폐기물 처리
Return refillable compressed gas cylinders to supplier. Incineration with added hydrocarbon fuel, controlled so as to produce elemental nitrogen, CO2, and water. Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/ mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal.참고 문헌
1.https://en.wikipedia.org/wiki/Nitric_oxide 2.https://www.drugs.com/mtm/nitric-oxide-inhalation-gas.html 3.https://www.britannica.com/science/nitric-oxide 4.http://pediatrics.aappublications.org/content/106/2/344 5.http://www.mensfitness.com/nutrition/supplements/supplement-guide-nitric-oxide 6.https://www.chemicalbook.com/productchemicalpropertiescb5433122.htm산화 질소 준비 용품 및 원자재
원자재
준비 용품
산화 질소 공급 업체
글로벌( 55)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-029-89586680 +86-18192503167 |
1026@dideu.com | China | 9358 | 58 |
| Mainchem Co., Ltd. | +86-0592-6210733 |
sale@mainchem.com | China | 32360 | 55 |
| Central China Special Gas (CCSG) Co., Ltd | 0734-8755555 15674722888 |
lyq@ccsg.cn | China | 281 | 58 |
| Guangzhou Yuejia Gas Co., Ltd | 400-6377517 19876107228 |
linfeng@yigas.cn | China | 39 | 58 |
| Shangchem Co., Ltd. | +86-21-68182121 |
China | 882 | 52 | |
| skychemical Co.Ltd | 021-20958197 20965099 |
sales@skychemical.com | China | 1184 | 57 |
| Beijing HuaMeiHuLiBiological Chemical | 010-56205725 |
waley188@sohu.com | China | 12338 | 58 |
| Shanghai T&W Pharmaceutical Co., Ltd. | +86 21 61551611 |
China | 9901 | 58 |
산화 질소 관련 검색:
이산화질소 질소 산화 질소
Nitrogen Tetroxide
NITROUS OXIDE
Nitrogen pentoxide
NEUTRALIZATION SOLUTION
DI-TERT-BUTYL NITROXIDE
NOS-1(bNOS:neuronal NOS:nNOS:Nitric Oxide synthase-1)
NIPECOTIC ACID
NOS-3 (eNOS) endotheli cell NOS
NOS-2 (iNOS) Nitric Oxide Synthase,Inducible
NITRIC OXIDE SYNTHASE (NEURONAL),NITRIC OXIDE SYNTHASE, NEURONAL RAT,RAT BRAIN NITRIC OXIDE SYNTHASE
Nitric oxide and dinitrogen tetroxide mixtures
NITRIC-15N OXIDE
NITRIC-15N OXIDE-18O
UNIVERSAL NITRIC OXIDE SYNTHASE (AB-2) MONOCLONAL ANTIBODY
ZINC NITROPRUSSIDE