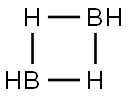디보란
|
|
디보란 속성
- 녹는점
- -165°C
- 끓는 점
- -93°C
- 밀도
- 0.477
- 인화점
- -90°C
- 용해도
- reacts with H2O
- 물리적 상태
- 무색 가스
- 색상
- colorless gas; flammable
- 냄새
- 1.8~3.5ppm에서 불쾌한 냄새 감지 가능
- 수용성
- 물에서 분해되다
- Merck
- 13,3039
- 노출 한도
- TLV-TWA 0.11 mg/m3 (0.1 ppm) (ACGIH and OSHA); IDLH 40 ppm (NIOSH).
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | F+;T,T,F+,T+ | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 12-23/24/25-36/37/38-26 | ||
| 안전지침서 | 9-16-36/37/39-45-36/37-28 | ||
| 유엔번호(UN No.) | UN 1911/1953 | ||
| 자연 발화 온도 | 38 to 52 °C | ||
| DOT ClassificationII | 2.3, Hazard Zone A (Gas poisonous by inhalation) | ||
| 위험 등급 | 2.3 | ||
| 유해 물질 데이터 | 19287-45-7(Hazardous Substances Data) | ||
| 독성 | LC50 inhal (rat) 50 ppm (4 h) PEL (OSHA) 0.1 ppm TLV-TWA (ACGIH) 0.1 ppm |
||
| IDLA | 15 ppm | ||
| 기존화학 물질 | KE-09913 | ||
| 사고대비 물질 필터링 | 56 |
디보란 C화학적 특성, 용도, 생산
물성
다이보레인(diborane, 디보란)은 B2H6 화학식을 갖는, 붕소와 수소를 이루는 화합물이다. 실내 온도에서는 달콤한 향의 무색의 기체이다. 다이보레인은 공기와 잘 섞이기 때문에 폭발 혼합물을 만들기 쉽다. 다이보레인은 실내 온도에서 습기가 찬 공기에 닿으면 자연 발화한다.존재
다이보레인은 19세기 금속 붕소화물을 가소분해하면서 처음 합성하였으나, 이에 대해 분석되지는 않았다. 1912년부터 1936년까지 앨프리드 스톡은 연구를 통해 에테인과 같은 구조의 다이보레인을 처음 제안하였다S. H. 바우어는 전자회절 측정을 통해 그가 제안한 구조를 지지하는 것으로 보였다.용도
디보란은 보란류의 하나로 고급 수소화붕소의 출발 물질로 중요한 화합물입니다.개요
Diborane is a colorless gas at room temperature and atmospheric pressure. It has an unpleasant, distinctive, sickly sweet odor. Diborane is a highly flammable gas that forms a flammable mixture with air over a range of 0.9 percent to 98 percent diborane (at 1 atm). Diborane bums in air (or oxygen) with a blue to green flame. Diborane is considered pyrophoric at room temperature. The gas is easily ignited by a spark or the heat of reaction with moisture in air.Pure diborane is insensitive to mechanical shock; however, shock and thermally sensitive mixtures may be formed in the presence of impurities such as oxygen, water, halogenated hydrocarbons, and so on. Thermal decomposition of diborane to hydrogen can result in excessive pressure buildup. Vessels for containment of diborane should be designed to contain such resultant decomposition pressure.
화학적 성질
Diborane is a compressed, colorless, and flam- mable gas. It has a nauseating, sickly sweet odor.용도
Diborane is used as a rocket propellant, in thevulcanization of rubber, as a polymerizationcatalyst, as a reducing agent, in the synthesisof trialkyl boranes, and as a doping agent(Merck 1996).공기와 물의 반응
Highly flammable. Ignites spontaneously in moist air (forms hydrogen and boric acid), [Haz. Chem. Data (1966)]. Oxygen and Diborane form spontaneously explosive mixtures, [J. Amer. Chem. Soc. 76, 1997(1954)].반응 프로필
Diborane is a colorless, air and moisture-sensitive gas, highly toxic. Diborane ignites in air. Diborane is very explosive when exposed to heat or flame, on contact with moisture Diborane produces hydrogen gas. Explosive reaction with benzene vapor, chlorine, nitric acid and tetravinyllead [Bretherick, 5th ed., 1995, p. 77]. Explosive reaction with dimethyl sulfoxide [Shriver, 1969, p. 209], violent reaction with halocarbon liquids used as fire extinguishants (e.g., carbon tetrachloride). Reaction with Al or Li produces complex hydrides that may ignite spontaneously in air [Haz. Chem. Data, 1975, p. 114].위험도
Diborane is pyrophoric and will ignite upon exposure to air. The boiling point is ?135°F and the flammable range is 0.8%–88% in air. The ignition temperature is 100° (37°C) to 140°F (60°C), and the flash point is 130°F (54°C). Diborane will react violently with halogenated fire-extinguishing agents, such as the halons. The four-digit UN identification number is 1911. The NFPA 704 designation is health 4, flammability 4, and reactivity 3. The white section of the diamond has a W with a slash through it, indicating water reactivity.건강위험
Boranes are highly toxic by inhalation, skin absorption or ingestion. They may produce acute or chronic poisoning. Diborane is an irritant to the lungs and kidneys. The primary effect of Diborane poisoning is lung congestion caused by local tissue irritation produced by the exothermic reaction of hydrolysis.화재위험
Diborane will ignite spontaneously in moist air at room temperature. Also, Diborane reacts violently with vaporizing liquid-type extinguishing agents. Diborane hydrolyzes in water to hydrogen and boric acid. Incompatible with air, halogenated compounds, aluminum, lithium, active metals, oxidized surfaces, chlorine, fuming nitric acid, nitrogen trifluoride, oxygen, and phosphorus trifluoride. Avoid moist air, electrical sparks, open flames or any other heat source. Hazardous polymerization may occur.인화성 및 폭발성
Diborane is a flammable gas that ignites spontaneously in moist air at room temperature and forms explosive mixtures with air from 0.8% up to 88% by volume. Diborane reacts with halogenated hydrocarbons, and fire extinguishing agents such as Halon or carbon tetrachloride are therefore not recommended. Carbon dioxide extinguishers should be used to fight diborane fires. Fires involving diborane sometimes release toxic gases such as boron oxide smoke.Materials Uses
Common metals are suitable as materials of construction. These include the following metals and metal alloys: chrome-molybdenum steel, Type 300 stainless steel, brass, lead, Monel, K-Monel, and nickel. Piping and appurtenances for undiluted diborane must be designed by experienced engineers and safety and fire protection specialists. Saran, polyethylene, Kel-F, Teflon, graphite, and high-vacuum silicone grease are satisfactory for use with diborane.In addition to the ability of a material to withstand chemical attack, the evaluation of materials compatibility with diborane should also emphasize the effect of the material on diborane stability (as expressed by the decomposition rate). The use of the following materials is not recommended:
? Metal oxides
? Natural rubbers
? Neoprene
? Leak-lock
? Permatex
? Ordinary oil and grease
? Nordel 1145 RPT elastomer, unfilled and Si02-filled
? silicon elastomer, unfilled and SiOrfilled
? CIS-4 polybutadiene elastomer, unfilled and SiOrfilled
잠재적 노출
Diborane is used as the source of boron in the semiconductor industry; as a catalyst for olefin polymerization; a rubber vulcanizer; a reducing agent; a flame-speed accelerator; a chemical intermediate for other boron hydrides; as a doping agent; in rocket propel- lants, and in the conversion of olefins to trialkyl boranes and primary alcohols.저장
diborane should be used only in a fume hood free of ignition sources and should be stored in a cold, dry, wellventilated area separated from incompatible substances and isolated from sources of sparks and open flames.운송 방법
UN1911 Diborane, Hazard Class: 2.3; Labels: 2.3-Poisonous gas, 2.1-Flammable gas Inhalation Hazard Zone A. Cylinders must be transported in a secure upright position, in a well-ventilated truck. Protect cylinder and labels from physical damage. The owner of the compressed gas cylinder is the only entity allowed by federal law (49CFR) to transport and refill them. It is a violation of transportation regulations to refill compressed gas cylinders without the express written permission of the owner.비 호환성
A strong reducing agent. Unstable above 8 C. The presence of contaminants may lower the autoigni- tion temperature; ignition may take place at, or below, room temperature. Diborane can polymerize, forming liquid pentaborane (See P:0190). It ignites spontaneously in moist air; and on contact with water, hydrolyzes exothermically forming hydrogen and boric acid. Contact with halogenated compounds (including fire extinguishers) may cause fire and explosion. Contact with aluminum, lithium and other active metals form hydrides which may ignite spontane- ously. Incompatible with aluminum, carbon tetrachloride; nitric acid; nitrogen trifluoride and many other chemicals. Reacts with oxidized surfaces. Attacks some plastics, rubber or coatings.폐기물 처리
Return refillable compressed gas cylinders to supplier. Incineration with aqueous scrub- bing of exhaust gases to remove B2O3 particulates.디보란 준비 용품 및 원자재
원자재
준비 용품
붕소
데노파민
2-(1-HYDROXYLAMINOETHYL)-BENZOTHIOPHENE
플루옥세틴
Ambroxane
CILAZAPRIL
HALOFANTRINE
Germane, bromo-(6CI,7CI,8CI,9CI)
1,1,2-Trimethylpropylborane
TRIPHENYLTIN HYDRIDE
BORANE-DIISOPROPYLAMINE
Dicyclohexylborane
Borane - 5-Ethyl-2-Methylpyridine CoMplex
Trimethoxyboroxine
9-BBN DIMER
트리메틸아민보란
BORANE-N,N-DIETHYLANILINE COMPLEX
Borane-triethylamine complex
디보란 관련 검색:
디보란 나트륨 붕소수화물 란타넘 보라이드(란탄 보리드) m-카르보레인 탄화 붕소 세륨 보리드 트리에틸보란 알루미늄 도데카보리드
BORON SILICIDE
NEODYMIUM BORIDE
BORON SILICIDE
PRASEODYMIUM BORIDE
YTTERBIUM BORIDE
TERBIUM BORIDE
HAFNIUM BORIDE
L-Threonyl-L-histidyl-L-threonyl-L-asparaginyl-L-isoleucyl-L-seryl-L-alpha-glutamyl-L-seryl-L-histidyl-L-prolyl-L-asparaginyl-L-alanyl-L-threonyl-L-phenylalanine
BIS(CATECHOLATO)DIBORANE
Bis(pinacolato)diboron