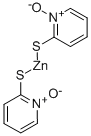
아연 피리치온 C화학적 특성, 용도, 생산
개요
Zinc pyrithione is a coordination complex of zinc and pyrithione that has antimicrobial and anticancer activities. It is active against the bacteria
E. coli,
S. aureus,
K. pneumoniae,
A. baumannii,
P. aeruginosa,
E. faecium,
E. faecalis, and
E. cloacae (MICs = 1-4 μg/ml) and the fungus
P. ovale when used at concentrations ranging from 0.01 to 10 μg/ml. Zinc pyrithione reduces tumor growth in an SCC-4 mouse xenograft model when administered at a dose of 1 mg/kg per week for six weeks. Formulations containing zinc pyrithione have been used in the treatment of dandruff.
화학적 성질
Beige granules
용도
Zinc pyrithione is directly cytotoxic and has antimicrobial effects. Zinc pyrithione is found in many shampoos (DHS-Zinc, Head and Shoulders) and should be applied for 5 minutes daily for 2 weeks.
Zinc pyrithione is the active ingredient in several shampoos used to control dandruff and seborrheic dermatitis and is also effective in the therapy of tinea versicolor. It remains unclear whether the beneficial effects are caused by an antiproliferative or antimicrobial effect or both. It is substantive to the hair, allowing continued therapeutic effect after washing.
Indications
Zinc pyrithione is the active ingredient in several shampoos used to control
dandruff and seborrheic dermatitis and is also effective in the therapy of tinea
versicolor. It remains unclear whether the beneficial effects are caused by an
antiproliferative or antimicrobial effect or both. It is substantive to the hair,
allowing continued therapeutic effect after washing.
일반 설명
Fine beige granules.
공기와 물의 반응
Insoluble in water.
반응 프로필
Organometallics, such as Zinc pyrithione, are reactive with many other groups. Incompatible with acids and bases. Organometallics are good reducing agents and therefore incompatible with oxidizing agents. Often reactive with water to generate toxic or flammable gases.
화재위험
Flash point data for Zinc pyrithione are not available, but Zinc pyrithione is probably combustible.
색상 색인 번호
Zinc pyrithione is widely used in antidandruff
shampoos and is a classic allergen. Concomitant reactions
are expected to both zinc and sodium pyrithione.
Safety Profile
Poison by ingestion,
skin contact, intraperitoneal, and
intravenous routes. Moderately toxic by
subcutaneous route. An experimental
teratogen. Experimental reproductive
effects. An eye irritant. When heated to
decomposition it emits very toxic fumes of
NOx, SOx, and ZnO. Used as an anti-
dandruff agent in shampoos. See also ZINC
COMPOUNDS and SULFIDES.
Synthesis
1000 g of sodium pyrithione, 20% aqueous solution (1.34 mmol of sodium pyrithione) was added to a 2L reactor equipped with a mechanical agitator and warmed to 70 °C with stirring. To the solution was added 190 g of 50 % zinc chloride (0.7 mmol of zinc chloride) over 10 minutes. After the completion of the addition, the solution was stirred at 70 °C for one hour, dehydrated and then washed with 500 g of distilled water. The solid thus obtained was dried with a heated-air drier at 70 °C to have a moisture content of less than 0.5 % and ground with a hammer mill to obtain a find powder of zinc pyrithione. The zinc pyrithione thus obtained was 212 g with a yield of 99.5 %.
아연 피리치온 준비 용품 및 원자재
원자재
준비 용품