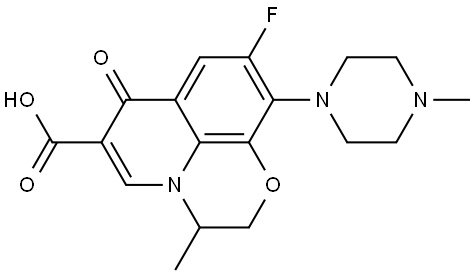
Ofloxacin
|
|
Ofloxacin 속성
- 녹는점
- 270-2750C
- 끓는 점
- 571.5±50.0 °C(Predicted)
- 밀도
- 1.2688 (estimate)
- 저장 조건
- Keep in dark place,Sealed in dry,Store in freezer, under -20°C
- 용해도
- 1M NaOH: 용해성50mg/mL
- 산도 계수 (pKa)
- 5.19±0.40(Predicted)
- 색상
- 에탄올에서 결정화 된 무색 바늘 모양의 물질
- 수용성
- 아세트산이나 물에 용해됩니다. 메탄올에 약간 용해됨
- 최대 파장(λmax)
- 326nm(H2O)(lit.)
- Merck
- 14,6771
- BCS Class
- 1
- CAS 데이터베이스
- 82419-36-1(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xn,Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 22-42/43-68-36/37/38 | ||
| 안전지침서 | 26-36/37/39-24/25-22-37/39-60 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | UU8815550 | ||
| HS 번호 | 29349990 | ||
| 유해 물질 데이터 | 82419-36-1(Hazardous Substances Data) | ||
| 독성 | LD50 in male, female mice, male, female rats (mg/kg): 5450, 5290, 3590, 3750 orally; 208, 233, 273, 276 i.v.; >10000, >10000, 7070, 9000 s.c. (Ohno) | ||
| 기존화학 물질 | KE-17021 |
Ofloxacin C화학적 특성, 용도, 생산
개요
Ofloxacin is an antibacterial agent with increased potency in comparison to the prototype third generation quinolone, norfloxacin. It has a broad spectrum of activity against gram-positive and gram-negative bacteria and is useful in the treatment of kidney, genitourinary, and upper respiratory tract infections.화학적 성질
Off-White Solid용도
Ofloxacin is a fluorinated quinolone antibacterial.This compound is a contaminant of emerging concern (CECs).Indications
Ofloxacin also possesses a broad spectrum of antimicrobial action. It is highly active with respect to Gram-negative microorganisms, such as blue-pus bacillus, hemophilic and colon bacillus, shigella, salmonella, and chlamydia. It is used for infections of the respiratory tract, ears, throat, nose, skin, soft tissue, bones, joints, infective-inflammatory diseases of the abdominal cavity organs (kidneys, urinary tract), and organs of the pelvis minor (genitalia), and for gonorrhea. Synonyms of this drug are tarivid, flobacin, and others.정의
ChEBI: An oxazinoquinolone carrying carboxy, fluoro, methyl and 4-methylpiperazino substituents. A synthetic fluoroquinolone antibacterial agent, it inhibits the supercoiling activity of bacterial DNA gyrase, halting DNA replication.Antimicrobial activity
It exhibits good activity against a wide range of enterobacteria, including strains resistant to nalidixic acid, as well as against Aeromonas, Campylobacter, Vibrio and Moraxella spp. Activity against methicillin-sensitive Staph. aureus is good, but streptococci, including Str. pneumoniae and enterococci, are less susceptible. Most anaerobes are moderately or completely resistant. It is active against L. pneumophila, Ch. pneumoniae, C. trachomatis, mycoplasmas, ureaplasmas and M. tuberculosis. Other mycobacteria such as M. fortuitum, M. kansasii, M. chelonei and the M. avium complex are moderately susceptible.Pharmaceutical Applications
A tricyclic 6-fluoro, 7-piperazinyl quinoline with a methyl substituted oxazine ring substituted. It is a racemic mixture of l- (levofloxacin, see p. 319) and d-isomers.Biotechnological Applications
Ofloxacin is a fluoroquinolone antibiotic present as a racemic mixture. Levofloxacin, S-isomer of ofloxacin, shows a broad spectrum of antibacterial activity against both gram-positive and gram-negative bacteria. The antibacterial activity of levofloxacin is 8–128 times greater than that of the corresponding R-isomer. A novel esterase of type B1 carboxylesterase/lipase family from a marine isolate Y. lipolytica CL180 was used to resolve a racemic mixture of ofloxacin ester. This esterase showed an enantioselectivity toward R, S-ofloxacin ester, and levofloxacin was produced with an enantiomeric excess of 52 % (Kim et al. 2007).Clinical Use
9-Fluoro-2,3-dihydro-3-methyl-10(4-methyl-1-piperazin-yl)-7-oxo-7H-pyrido[1,2,3-de]-1,4,-benzoxazine-6-carboxylicacid (Floxin, Floxin IV) is a member of the quinolone class of antibacterial drugs wherein the 1- and 8-positions are joined in the form of a 1,4-oxazine ring.Ofloxacin has been approved for the treatment of infectionsof the lower respiratory tract, including chronic bronchitisand pneumonia, caused by Gram-negative bacilli. It isalso used for the treatment of pelvic inflammatory diseaseand is highly active against both gonococci and chlamydia.In common with other fluoroquinolones, ofloxacin is not effectivein the treatment of syphilis. A single 400-mg oraldose of ofloxacin in combination with the tetracycline antibioticdoxycycline is recommended by the Centers forDisease Control and Prevention (CDC) for the outpatienttreatment of acute gonococcal urethritis. Ofloxacin is alsoused for the treatment of urinary tract infections caused byGram-negative bacilli and for prostatitis caused by E. coli.Infections of the skin and soft tissues caused by staphylococci,streptococci, and Gram-negative bacilli may also betreated with ofloxacin.
부작용
Untoward reactions havebeen described in 2.5–7.5% of patients, and are those common to the group: gastrointestinal tract disturbances, rashes, tendon rupture and insomnia. CNS effects rarely occur.Safety Profile
Poison by intravenous route.Moderately toxic by ingestion. An experimental teratogen.Other experimental reproductive effects. Human systemiceffects: body temperature increase, diarrhea,hallucinations, hypermotility, irritability, psychosis.Mutation dOfloxacin 준비 용품 및 원자재
원자재
수소화나트륨
DE
프탈이미드
Ethyl 2,3,4,5-tetrafluorobenzoyl acetate
2,3,4,5-사불화 벤조익산
트리플르오르-1,2,3,-4니트로벤젠
다이메틸 설폭사이드
N-메틸피페라진
에톡시메틸렌말론산 디에틸에스테르
히드로아황산 나트륨
준비 용품
Ofloxacin 공급 업체
글로벌( 539)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| XINGTAI XINGJIU NEW MATERIAL TECHNOLOGY CO., LTD | +8617731987558 |
xingjiu@xingjiubiotech.com | China | 990 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12456 | 58 |
| Shaanxi TNJONE Pharmaceutical Co., Ltd | +8618740459177 |
sarah@tnjone.com | China | 874 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29797 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| Hubei XinRunde Chemical Co., Ltd. | +8615102730682 |
bruce@xrdchem.cn | CHINA | 566 | 55 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| SHANDONG ZHI SHANG CHEMICAL CO.LTD | +86 18953170293 |
sales@sdzschem.com | China | 2931 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 |
alice@crovellbio.com | China | 8823 | 58 |