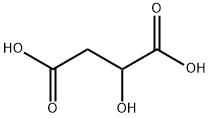
말레익산
|
|
말레익산 속성
- 녹는점
- 131-133 °C (lit.)
- 끓는 점
- 167.16°C (rough estimate)
- 알파
- [α]D20 -0.5~+0.5° (c=5, H2O)
- 밀도
- 1.609
- 증기 밀도
- 4.6 (vs air)
- 증기압
- <0.1 mm Hg ( 20 °C)
- 굴절률
- 1.3920 (estimate)
- 인화점
- 203 °C
- 저장 조건
- 2-8°C
- 용해도
- 메탄올: 0.1 g/mL, 투명, 무색
- 산도 계수 (pKa)
- 3.4(at 25℃)
- 색상
- 흰색에서 거의 흰색
- 냄새
- 100.00%에서. 냄새 없는
- 수소이온지수(pH)
- 3.33(1 mM solution);2.74(10 mM solution);2.21(100 mM solution);
- optical activity
- [α]/D 0.10 to +0.10°
- ?? ??
- 냄새 없는
- 수용성
- 558g/L(20℃)
- Merck
- 14,5707
- BRN
- 1723539
- LogP
- -6.14 at 20℃
- CAS 데이터베이스
- 6915-15-7(CAS DataBase Reference)
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xn,Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 22-37/38-41-36/37/38-42/43-34 | ||
| 안전지침서 | 26-39-37/39-36-36/39 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | ON7175000 | ||
| 자연 발화 온도 | 644 °F | ||
| TSCA | Yes | ||
| HS 번호 | 29181980 | ||
| 유해 물질 데이터 | 6915-15-7(Hazardous Substances Data) | ||
| 독성 | LD50 orally in Rabbit: > 3200 mg/kg | ||
| 기존화학 물질 | KE-20414 |
말레익산 C화학적 특성, 용도, 생산
개요
사과산은 사과등 천연과일 및 야채에 다량 함유되어 있으며, 사람의 탄수화물 신진대사에 필요한 산미제입니다. 같은 산미제인 구연산에 비해 산도가 강하며 사과산을 사용할 경우 약 20%가 절약될 뿐만 아니라 우아하고 세련된 고급 산미를 나타내고 산미(酸味) 지속이 훨씬 길어 사람의 입맛을 한층 즐겁게 하여 줍니다.용도
특히 구미에서는 Aspartame등 인조당을 사용하는 건강식품(Diet Food/Drinks)제조에는 필수불가결하게 사용됩니다. 또한사과산은 융점이 낮아 캔디(Candy)등 딱딱한 식품의 산미제로도 많이 쓰입니다.순도시험
(1) 융점 : 이 품목의 융점은 127~132℃이어야 한다.
(2) 용상 : 이 품목 1g을 물 20mL에 녹일 때, 그 액은 징명하여야 한다.
(3) 염화물 : 이 품목 1g을 취하여 염화물시험법에 따라 시험할 때, 그 양은 0.01N 염산 0.1mL에 대응하는 양 이하이어야 한다.
(4) 비소 : 이 품목을 비소시험법에 따라 시험할 때, 그 양은 4.0ppm 이하이어야 한다.
(5) 납 : 이 품목 5.0g을 취하여 원자흡광광도법 또는 유도결합플라즈마발광광도법에 따라 시험할 때, 그 양은 2.0ppm 이하이어야 한다.
(6) 수은 : 이 품목을 수은시험법에 따라 시험할 때, 그 양은 1.0ppm 이하이어야 한다.
(7) 산화되기 쉬운 물질 : 이 품목 0.1g을 취하여 물 25mL 및 황산(1→20) 25mL를 가하여 녹인 다음 이를 20℃로 유지하고 0.1N 과망간산칼륨용액 1mL를 가할 때, 액의 홍색은 3분 이내에 없어져서는 아니 된다.
확인시험
(1) 이 품목의 수용액(1→20) 1mL를 시험관에 취하고 레소르신 2~3mg 및 황산 1mL를 가하여 흔들어 섞고 120~130℃로 5분간 가열하여 식힌 다음 물을 가하여 5mL로 한다. 이에 식히면서 수산화나트륨용액(2→5)을 가하여 알칼리성으로 하고 다시 물을 가하여 10mL로 하면 이 액은 자외선 아래에서 엷은 청색의 형광을 나타낸다.
(2) 이 품목의 수용액(1→20)을 자제접시에 취하고 암모니아시액으로 중화한 다음 설파닐산 10mg을 가하여 수욕상에서 수분간 가열하고 다시 아질산나트륨용액(1→5) 5mL를 가하여 조금 가온한 다음 수산화나트륨시액으로 알칼리성으로 하면 적색을 나타낸다.
정량법
이 품목 1.5g을 정밀히 달아 물에 녹여 250mL로 하고 그 중 25mL를 취하여 0.1N 수산화나트륨용액으로 적정한다(지시약 : 페놀프탈레인시액 2방울).
0.1N 수산화나트륨용액 1mL = 6.704mg C4H6O5
강열잔류물
이 품목의 강열잔류물은 0.05% 이하이어야 한다.
용도
Malic acid, HOOCCH(OH).CH2COOH, also known as hydroxysuccinic acid, is a colorless solid. It is soluble in water and alcohol. Malic acid exists in two optically active forms and a racemic mixture. It is used in medicine and found in apples and other fruits.The naturally occuring isomer is the L-form which has been found in apples and many other fruits and plants. Selective α-amino protecting reagent for amino acid derivatives. Versatile synthon for the preparation of chiral compounds including κ-opioid rece
정의
ChEBI: Malic acid is a 2-hydroxydicarboxylic acid that is succinic acid in which one of the hydrogens attached to a carbon is replaced by a hydroxy group. It has a role as a food acidity regulator and a fundamental metabolite. It is a 2-hydroxydicarboxylic acid and a C4-dicarboxylic acid. It derives from a succinic acid. It is a conjugate acid of a malate(2-) and a malate.생산 방법
Malic acid is manufactured by hydrating maleic and fumaric acids in the presence of suitable catalysts. The malic acid formed is then separated from the equilibrium product mixture.일반 설명
The chiral resolution of DL-malic acid by ligand-exchange capillary electrophoresis was studied.Pharmaceutical Applications
Malic acid is used in pharmaceutical formulations as a generalpurpose acidulant. It possesses a slight apple flavor and is used as a flavoring agent to mask bitter tastes and provide tartness. Malic acid is also used as an alternative to citric acid in effervescent powders, mouthwashes, and tooth-cleaning tablets.In addition, malic acid has chelating and antioxidant properties. It may be used with butylated hydroxytoluene as a synergist in order to retard oxidation in vegetable oils. In food products it may be used in concentrations up to 420 ppm.
Therapeutically, malic acid has been used topically in combination with benzoic acid and salicylic acid to treat burns, ulcers, and wounds. It has also been used orally and parenterally, either intravenously or intramuscularly, in the treatment of liver disorders, and as a sialagogue.
Mechanism of action
Malic acid is absorbed from the gastrointestinal tract from whence it is transported via the portal circulation to the liver. There are a few enzymes that metabolize malic acid. Malic enzyme catalyzes the oxidative decarboxylation of L-malate to pyruvate with concomitant reduction of the cofactor NAD+ (oxidized form of nicotinamide adenine dinucleotide) or NADP+ (oxidized form of nicotinamide adenine dinucleotide phosphate). These reactions require the divalent cations magnesium or manganese. Three isoforms of malic enzyme have been identified in mammals: a cytosolic NADP+-dependent malic enzyme, a mitochondrial NADP+- dependent malic enzyme and a mitochondrial NAD(P)+-dependent malic enzyme. The latter can use either NAD+ or NADP+ as the cofactor but prefers NAD+. Pyruvate formed from malate can itself be metabolized in a number of ways, including metabolism via a number of metabolic steps to glucose. Malate can also be metabolized to oxaloacetate via the citric acid cycle. The mitochondrial malic enzyme, particularly in brain cells may play a key role in the pyruvate recycling pathway, which utilizes dicarboxylic acids and substrates, such as glutamine, to provide pyruvate to maintain the citric acid cycle activity when glucose and lactate are low.Safety Profile
A poison by intraperitoneal route. Moderately toxic by ingestion. A skin and severe eye irritant. When heated to decomposition it emits acrid smoke and irritating fumes.Safety
Malic acid is used in oral, topical, and parenteral pharmaceutical formulations in addition to food products, and is generally regarded as a relatively nontoxic and nonirritant material. However, concentrated solutions may be irritant.LD50 (rat, oral): 1.6 g/kg(3)
LD50 (rat, IP): 0.1 g/kg
저장
Malic acid is stable at temperatures up to 150°C. At temperatures above 150°C it begins to lose water very slowly to yield fumaric acid; complete decomposition occurs at about 180°C to give fumaric acid and maleic anhydride.Malic acid is readily degraded by many aerobic and anaerobic microorganisms. Conditions of high humidity and elevated temperatures should be avoided to prevent caking.
The effects of grinding and humidity on malic acid have also been investigated.
The bulk material should be stored in a well-closed container, in a cool, dry place.
Purification Methods
Crystallise the acid from acetone, then from acetone/CCl4, or from ethyl acetate by adding pet ether (b 60-70o). Dry it at 35o under 1mm pressure to avoid formation of the anhydride. [Beilstein 3 IV 1124.]비 호환성
Malic acid can react with oxidizing materials. Aqueous solutions are mildly corrosive to carbon steels.Regulatory Status
GRAS listed. Both the racemic mixture and the levorotatory isomer are accepted as food additives in Europe. The DL and L forms are included in the FDA Inactive Ingredients Database (oral preparations). Included in nonparenteral and parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Nonmedicinal Ingredients.말레익산 준비 용품 및 원자재
원자재
준비 용품
말레익산 공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Dideu Industries Group Limited | +86-29-89586680 +86-15129568250 |
1026@dideu.com | China | 29271 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12456 | 58 |
| Hebei Yime New Material Technology Co., Ltd. | +86-66697723 +86-17703311139 |
admin@china-yime.com | China | 563 | 58 |
| Hebei Jingbo New Material Technology Co., Ltd | +8619931165850 |
hbjbtech@163.com | China | 1000 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 |
info@fdachem.com | China | 7786 | 58 |
| Hebei Saisier Technology Co., LTD | +86-18400010335 +86-13102810335 |
admin@hbsaisier.cn | China | 690 | 58 |
| Ouhuang Engineering Materials (Hubei) Co., Ltd | +8617702722807 |
admin@hbouhuang.com | China | 1696 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29797 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| Shanghai Time Chemicals CO., Ltd. | +86-021-57951555 +8617317452075 |
jack.li@time-chemicals.com | China | 1807 | 55 |