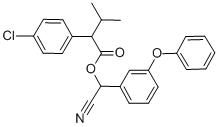
펜발러레이트
|
|
펜발러레이트 속성
- 녹는점
- 54-59℃
- 끓는 점
- 300°C
- 밀도
- d23 1.17
- 증기압
- 1.92×10-5 Pa (20 °C)
- 굴절률
- nD20 1.5533
- 저장 조건
- Sealed in dry,2-8°C
- 용해도
- DMSO:1.0(Max Conc. mg/mL);2.38(Max Conc. mM)
DMF:30.0(Max Conc. mg/mL);71.44(Max Conc. mM)
Ethanol:5.0(Max Conc. mg/mL);11.91(Max Conc. mM)
- 수용성
- 20°C에서 100% 에탄올(>25mg/ml), 물(0.001g/L) 및 DMSO에 용해됩니다.
- Merck
- 13,4038
- BRN
- 2025982
- IARC
- 3 (Vol. 53) 1991
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | T,N | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 25-36/37/38-50/53-57 | ||
| 안전지침서 | 26-45-60-61 | ||
| 유엔번호(UN No.) | UN 2811 6.1/PG 3 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | CY1576350 | ||
| 위험 등급 | 6.1(b) | ||
| 포장분류 | III | ||
| HS 번호 | 29269090 | ||
| 유해 물질 데이터 | 51630-58-1(Hazardous Substances Data) | ||
| 독성 | LD50 orally in rats: 451 mg/kg (DMSO); >3200 mg/kg (aqueous suspension), Shell Technical Data Bulletin | ||
| 기존화학 물질 | KE-05733 | ||
| 유해화학물질 필터링 | 97-1-336 | ||
| 함량 및 규제정보 | 물질구분: 유독물질; 혼합물(제품)함량정보: 펜발러레이트 및 이를 1% 이상 함유한 혼합물 |
| 그림문자(GHS): |
 
|
||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 신호 어: | Danger | ||||||||||||||||||||||||||||||||||||||||||
| 유해·위험 문구: |
|
||||||||||||||||||||||||||||||||||||||||||
| 예방조치문구: |
|
펜발러레이트 C화학적 특성, 용도, 생산
개요
Fenvalerate is a yellow-brown viscous liquid that is practically soluble in water. It is stable to moderate heat and light and is rapidly hydrolysed in basic environments (above pH 8). Fenvalerate is a racemic mixture of four stereoisomers in equal proportions owing to the presence of two chiral centres. It is a synthetic type II pyrethroid.Fenvalerate is registered as an insecticide for a wide array of crop uses, as well as a termiticide and insect repellent.
화학적 성질
It is an ester of 2-(4-chlorophenyl)-3-methylbutyric acid and alpha-cyano-3-phenoxybenzyl alcohol but lacks a cyclopropane ring. However, in terms of its insecticidal behaviour, it belongs to the pyrethroid insecticides. Technical grade fenvalerate is a yellow or brown viscous liquid having a specific gravity of 1.175 at 25°C. The vapour pressure is 0.037 mPa at 25°C, and it is relatively non-volatile. It is practically insoluble in water (approximately 2 μg/L) but soluble in organic solvents such as acetone, xylene, and kerosene. It is stable to light, heat, and moisture but unstable in alkaline media.용도
Fenvalerate controls a wide range of insect pests in fruit, vines, olives, hops, nuts, vegetables, cotton, oilseed rape, sunflowers, alfalfa, cereals, maize, sorghum, potatoes, beets, soyabeans, tobacco, sugar cane and ornamentals. It is also used in public health situations and in animal houses.정의
ChEBI: A carboxylic ester obtained by formal condensation between 2-(4-chlorophenyl)-3-methylbutyric acid and cyano(3-phenoxyphenyl)methanol.일반 설명
A clear viscious yellow liquid with a mild odor. Used as broad spectrum insecticide.공기와 물의 반응
Insoluble in water. Rapidly hydrolyzed by alkaline solution.반응 프로필
A pyrethroid. Phenvalerate is an ester and nitrile. Esters react with acids to liberate heat along with alcohols and acids. Strong oxidizing acids may cause a vigorous reaction that is sufficiently exothermic to ignite the reaction products. Heat is also generated by the interaction of esters with caustic solutions. Flammable hydrogen is generated by mixing esters with alkali metals and hydrides. Nitriles may polymerize in the presence of metals and some metal compounds. They are incompatible with acids; mixing nitriles with strong oxidizing acids can lead to extremely violent reactions. Nitriles are generally incompatible with other oxidizing agents such as peroxides and epoxides. The combination of bases and nitriles can produce hydrogen cyanide. Nitriles are hydrolyzed in both aqueous acid and base to give carboxylic acids (or salts of carboxylic acids).위험도
Questionable carcinogen.색상 색인 번호
Fenvalerate is an insecticide of the synthetic pyrethroid group, which induced sensitization in farmers.Safety Profile
Poison by ingestion, intravenous, and intracerebral routes. Moderately toxic by skin contact.Experimental reproductive effects. Mutation data reported. Highly toxic to fish and bees. Corrosive, causes eye damage. A skin irritant. When heated to decomposition it emits toxic fumes of Cl-, NOx, and CN-. See also CYANIDE.잠재적 노출
Fenvalerate is one of the most versatile synthetic pyrethroid insecticides. It is mostly used in agriculture and on cattle, but also in homes and gardens. It acts as a stomach poison against a wide variety of leaf and fruit eating such as bollworm fruit and shoot borers and aphids. Crops on which it is used include cotton, cauliflower, okra, vines and fruits. It is also used in public health and animal husbandry. It is effective against pests whose strains are resistant to organochlorine, organophosphorus, and carbamate insecticides. Not used in EU countries환경귀착
Soil. Fenvalerate is moderately persistent in soil. The percentage of the initial dosage (1 ppm) remaining after 8 weeks of incubation in an organic and mineral soil were 58 and 12%, respectively, while in sterilized controls 100 and 91% remained, respectively (Chapman et al., 1981).In a sugarcane runoff plot, fenvalerate was applied at a rate of 0.22 kg/ha 4 times each year in 1980 and 1981. Runoff losses in 1980 and 1981 were 0.08 and 0.56 of the applied amount, respectively (Smith et al., 1983).
Plant. Dislodgable residues of fenvalerate on cotton leaf 0, 24, 48, 72 and 96 hours after application (0.22 kg/ha) were 0.85, 0.36, 0.38, 0.28 and 0.28 μg/m2, respectively (Buck et al., 1980).
Surface Water. In an estuary, the half-life of fenvalerate was 27–42 days (Schimmel et al., 1983).
Chemical/Physical. Undergoes hydrolysis at the ester bond (Hartley and Kidd, 1987). Decomposes gradually at 150–300°C (Windholz et al., 1983) probably releasing toxic fumes of nitrogen and chlorine.
신진 대사 경로
After foliar treatment of 14C-fenvalerate on wheat plants, the amount of residual radioactivity in the grain and hull is below the limit of reliable measurement. Individual degradation products accounting for more than 1% of the applied radioactivity are not present in the foliage or straw. Important degradation pathways include decarboxylation and ester cleavage.운송 방법
UN3349 Pyrethroid pesticide, solid toxic, Hazard Class: 6.1; Labels: 6.1-Poisonous material. UN3352 Pyrethroid pesticide, liquid toxic, Hazard Class: 6.1; Labels: 6.1-Poisonous materials.비 호환성
ncompatible with oxidizers, chlorates nitrates, peroxides, sulfuric acid, caustics, ammonia, aliphatic amines, alkanolamines, isocyanates, alkylene oxides, epichlorohydrin. Moisture may cause hydrolysis or other forms of decomposition. Emulsifiable concentrate is corrosive폐기물 처리
Incineration would be an effective disposal procedure where permitted. If an efficient incinerator is not available, the product should be mixed with large amounts of combustible material and contact with the smoke should be avoided. In accordance with 40CFR165, follow recommendations for the disposal of pesticides and pesticide containers참고 문헌
[1] http://www.toxipedia.org[2] Y. Xia, Q. Bian, L. Xu, S. Cheng, L. Song, J. Liu, W. Wu, S. Wang and X. Wang, Genotoxic effects on human spermatozoa among pesticide factory workers exposed to fenvalerate, Toxicology, 2004, vol. 203, 49-60
[3] Terry R Roberts, David H Hutson, Philip W Lee, Peter H Nicholls and Jack R Plimmer, Metabolic Pathways of Agrochemicals: Part 2: Insecticides and Fungicides, 1999
펜발러레이트 준비 용품 및 원자재
원자재
헵테인
Isopropyl(4-chlorophenyl)acetyl chloride
3-페녹시벤즈알데하이드
염화벤질
이소프로필브로마이드
phenylacetonitrile
2-(4-CHLOROPHENYL)-3-METHYLBUTYRONITRILE
테트라부틸암모늄 브로마이드
p-클로로벤질 시아나이드
부탄 산
염화나트륨
2-(4-Chlorophenyl)-3-methylbutyric acid
톨루엔
포스포러스옥시클로라이드
부티릴클로라이드
인 펜타염화물
사이안화 나트륨
포스겐
4-클로로페닐초산
cumenesulphonic acid
수산화나트륨
3-PHENOXYPHENYLACETONITRILE
이소발레릴산
이소프로필알코올
Benzene
황산
이소발레로일클로라이드
클로로프로판(2-)
준비 용품
펜발러레이트 공급 업체
글로벌( 299)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Yanxi Chemical Co., Ltd. | +8617531190177 |
peter@yan-xi.com | China | 5993 | 58 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 |
fandachem@gmail.com | China | 9352 | 55 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-61398051 +8613650506873 |
sales@chemdad.com | China | 39916 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 |
sales@conier.com | China | 47465 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-29-87569266 15319487004 |
1015@dideu.com | China | 2263 | 58 |
| Zhengzhou Alfa Chemical Co.,Ltd | +8618530059196 |
sale04@alfachem.cn | China | 12431 | 58 |
| TargetMol Chemicals Inc. | +1-781-999-5354 +1-00000000000 |
marketing@targetmol.com | United States | 19892 | 58 |