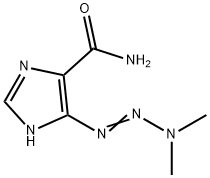
다카바진
|
|
다카바진 속성
- 녹는점
- 199-205°C
- 끓는 점
- 315.57°C (rough estimate)
- 밀도
- 1.3206 (rough estimate)
- 굴절률
- 1.7500 (estimate)
- 저장 조건
- 2-8°C
- 용해도
- 물과 무수 에탄올에 약간 용해되고 염화메틸렌에는 거의 용해되지 않습니다.
- 산도 계수 (pKa)
- pKa 4.42 (Uncertain)
- 물리적 상태
- 가루
- 색상
- 황백색에서 밝은 노란색까지
- 수용성
- Slightly soluble in water, DMSO and ethanol /n
- Merck
- 14,2798
- LogP
- -0.240
- CAS 데이터베이스
- 4342-03-4(CAS DataBase Reference)
- IARC
- 2B (Vol. 26, Sup 7) 1987
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | T,Xi,Xn | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 45-46-20/21/22-36/37/38 | ||
| 안전지침서 | 53-36/37/39-45-37/39-36-26 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | NI3950000 | ||
| HS 번호 | 29349990 | ||
| 유해 물질 데이터 | 4342-03-4(Hazardous Substances Data) |
다카바진 C화학적 특성, 용도, 생산
개요
Dacarbazine is nevertheless considered the first representative of the series of triazene derivatives. It has been shown that it is an alkylating agent, and thus this drug inhibits RNA and protein synthesis to a greater degree than DNA. Dacarbazine is used intravenously for Hodgkin’s disease, soft-tissue sarcoma, and metastatic melanoma. A synonym of this drug is diticene.화학적 성질
Dacarbazine is a white to ivory-colored crystalline solid.용도
Dacarbazine is used as an antineoplastic for treatment of malignant melanoma and sarcomas.정의
ChEBI: A monocarboxylic acid amide that is 1H-imidazole-4-carboxamide which is substituted at position 5 by a 3,3-dimethyltriaz-1-en-1-yl group. It is used for the treatment of metastatic malignant melanoma, and in combination with other drugs or the treatment of Hodgkin's disease and soft-tissue sarcoma.Indications
Dacarbazine (DTIC-Dome) is activated by photodecomposition and by enzymatic N-demethylation. Eventual formation of a methyl carbonium ion results in methylation of DNA and RNA and inhibition of nucleic acid and protein synthesis. As with other alkylating agents, cells in all phases of the cell cycle are susceptible to dacarbazine.The plasma half-life of dacarbazine is biphasic, with a distribution phase of 19 minutes and an elimination phase of 5 hours. The drug is not appreciably protein bound, and it does not enter the central nervous system (CNS). Urinary excretion of unchanged drug is by renal tubular secretion. Dacarbazine metabolism and decomposition is complex.
Dacarbazine is the most active agent used in metastatic melanoma, producing a 20% remission rate. It is also combined with doxorubicin and other agents in the treatment of various sarcomas and Hodgkin’s disease.
Dacarbazine may cause severe nausea and vomiting. Leukopenia and thrombocytopenia occur 2 weeks after treatment, with recovery by 3 to 4 weeks. Less common is a flulike syndrome of fever, myalgias, and malaise. Alopecia and transient abnormalities in renal and hepatic function also have been reported.
일반 설명
Dacarbazine is available in 100- and 200-mg vials for IVadministration in the treatment of Hodgkin’s disease, malignantmelanoma, carcinoid cancer, neuroblastoma, andsoft tissue sarcoma. Resistance to dacarbazine has been primarilyattributed to enhanced activity of AGAT. The volumeof distribution exceeds the amount of water in thebody suggesting the compound distributes into body tissuespossibly the liver. The agent is not highly protein bound(20%) and is metabolized in the liver by CYP to give MTICand 4-amino-5-imidazole-carboxamide (AIC).Thedemethylation reaction is mediated by isozymes CYP1A1/2and CYP2E1. Elimination occurs via the urine with 40% to50% occurring as unchanged drug. Dose-limiting myelosuppressionpresents as both leucopenia and thrombocytopenia.Other adverse effects include nausea, vomiting,flulike symptoms, photosensitivity, and pain at the injectionsite.공기와 물의 반응
Insoluble in water.반응 프로필
Dacarbazine decomposes explosively at its melting point (250°C). Decomposes in the presence of light. Sensitive to oxidation.화재위험
Flash point data for Dacarbazine are not available. Dacarbazine is probably combustible.Clinical Use
This DNA methylating agent is administered IV as a single agent in the treatment of malignant melanoma and in combination with other agents in the treatment of metastatic melanoma.부작용
Approximately 40% of the drug is excreted unchanged, but both the 5-aminoimidazole-4-carboxamide (AIC, formed through the action of CYP1A enzymes) and the carboxylic acid (AIC hydrolysis product) are major urinary metabolites. Leukopenia and thrombocytopenia are the most common side effects and may be fatal. Patients also are at risk for hepatotoxicity, including hepatocellular necrosis.Safety Profile
Confirmed carcinogen with experimental carcinogenic and tumorigenic data. Poison by intraperitoneal and parenteral routes. Moderately toxic by ingestion and intravenous routes. Experimental teratogenic effects. Human systemic effects by intravenous route: nausea or vomiting, leukopenia (reduced white blood cell count), and changes in dehydrogenase enzymatic activity. Mutation data reported. When heated to decomposition it emits toxic fumes of NOx.잠재적 노출
Dacarbazine is used in cancer chemo- therapy. Dacarbazine is used as an antineoplastic agent in the treatment of certain skin cancers, and is occasionally used in the therapy of other neoplastic diseases which have become resistant to alternative treatment.br Health professionals who handle this drug (for example, pharmacists, nurses, and physicians) may possibly be exposed during drug preparation, administration, or cleanup; however, the risks can be avoided through use of appropriate containment equipment and work practices .People receiving dacarbazine in treatment are also exposed.환경귀착
The exact mechanism of action of dacarbazine is unknown; however, several proposed mechanisms have been made including inhibition of DNA synthesis by acting as a purine analog, alkylating agent, and interference with sulfhydryl groups. It is most commonly classified as an alkylating agent in the triazene group. While the active compound of dacarbazine, DTIC, is structurally similar to purines, its primary mechanism of action precludes the agent from being classified as an antimetabolite. Dacarbazine is a synthetic compound that is metabolically activated to the active alkylating metabolite methyl-triazeno-imidazole-carboxamide (MTIC) via the cytochrome P450 system, primarily CYP1A1, CYP1A2, and CYP 2E1. MTIC is rapidly tautomerized into an inactive derivative, 5-aminoimidazole-4-carboxamide (AIC), which is renally excreted. The entire process of activating DTIC occurs within 15 min of intravenous infusion. DTIC exerts its actions throughout all phases of the cellular cycle. The antitumor effects of this compound are related to the induction of methyl adducts to DNA. The 70% of alkylation occurs at the N7 position of guanine. The cytotoxic and mutagenic effects of MTIC are manifested through alkylation of DNA at the O6 guanine position, accounting for 6–8% of methylated bases formed. This is primarily a result of generation of incorrect base pairing, leading to DNA double strand breaks and apoptosis.운송 방법
UN3249 Medicine, solid, toxic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials.비 호환성
ncompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explo- sions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, and epoxides. Explosive decom- position reported @ 250℃ 255℃폐기물 처리
It is inappropriate and possi- bly dangerous to the environment to dispose of expired or waste drugs and pharmaceuticals by flushing them down the toilet or discarding them to the trash. Household quanti- ties of expired or waste pharmaceuticals may be mixed with wet cat litter or coffee grounds, double-bagged in plastic, discard in trash. Larger quantities shall carefully take into consideration applicable DEA, EPA, and FDA regulations. If possible return the pharmaceutical to the manufacturer for proper disposal being careful to properly label and securely package the material. Alternatively, the waste pharmaceutical shall be labeled, securely packaged and transported by a state licensed medical waste contractor to dispose by burial in a licensed hazardous or toxic waste landfill or incinerator.다카바진 준비 용품 및 원자재
원자재
준비 용품
다카바진 공급 업체
글로벌( 339)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12457 | 58 |
| Beijing Cooperate Pharmaceutical Co.,Ltd | 010-60279497 |
sales01@cooperate-pharm.com | CHINA | 1811 | 55 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21695 | 55 |
| Shanghai Zheyan Biotech Co., Ltd. | 18017610038 |
zheyansh@163.com | CHINA | 3620 | 58 |
| SHANDONG ZHI SHANG CHEMICAL CO.LTD | +86 18953170293 |
sales@sdzschem.com | China | 2931 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 |
alice@crovellbio.com | China | 8823 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| BOC Sciences | +1-631-485-4226 |
inquiry@bocsci.com | United States | 19553 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 |
sales@conier.com | China | 47465 | 58 |
| career henan chemical co | +86-0371-86658258 15093356674; |
factory@coreychem.com | China | 29826 | 58 |
다카바진 관련 검색:
다이메틸 설폭사이드 N,N-다이메틸폼아마이드 에탄 디메틸 에테르 다이메틸 푸마르산 이미다졸 디메틸 카보네이트 디메틸설페이트 N,N-디메틸아세트아미드 다카바진
Nicarbazin
Dacthal
5-amino-4-imidazolecarboxamide hydrochloride (intermediate of dacarbazine)
1-TETRACENE
Dacarbazine-D6
Dacarbazine impurity A
DACARBAZINE RELATED COMPOUND B (50 MG) (2-AZAHYPOXANTHINE)
CARBOXAMIDE