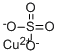황산동(II)
|
|
황산동(II) 속성
- 녹는점
- 200 °C (dec.)(lit.)
- 밀도
- 3.603 g/mL at 25 °C(lit.)
- 증기압
- 7.3 mm Hg ( 25 °C)
- 저장 조건
- Store at +5°C to +30°C.
- 용해도
- H2O: 용해성
- 물리적 상태
- 가루
- Specific Gravity
- 3.603
- 색상
- 약간 녹색을 띤 회색
- 수소이온지수(pH)
- 3.5-4.5 (50g/l, H2O, 20℃)
- 냄새
- 냄새 없는
- pH 범위
- 3.7 - 4.5
- 수용성
- 203g/L(20℃)
- 감도
- Hygroscopic
- Merck
- 14,2653
- Dielectric constant
- 10.3(0.0℃)
- 노출 한도
- ACGIH: TWA 1 mg/m3
NIOSH: IDLH 100 mg/m3; TWA 1 mg/m3
- 안정성
- 흡습성
- InChIKey
- ARUVKPQLZAKDPS-UHFFFAOYSA-L
- CAS 데이터베이스
- 7758-98-7(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xn,N,Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 36/38-50/53-22-51/53-36/37/38 | ||
| 안전지침서 | 24/25-36-60-61-22-26 | ||
| 유엔번호(UN No.) | UN 3288 6.1/PG 3 | ||
| WGK 독일 | 2 | ||
| RTECS 번호 | GL8800000 | ||
| F 고인화성물질 | 3 | ||
| TSCA | Yes | ||
| HS 번호 | 2833 25 00 | ||
| 위험 등급 | 6.1 | ||
| 포장분류 | III | ||
| 유해 물질 데이터 | 7758-98-7(Hazardous Substances Data) | ||
| 독성 | LD50 orally in Rabbit: 481 mg/kg | ||
| 기존화학 물질 | KE-08956 |
황산동(II) C화학적 특성, 용도, 생산
화학적 성질
Cupric sulfate, a bluish crystalline powder, also known as hydrocyanite and copper sulfate, vitriol, chalcanthite, and bluestone, is an azure blue material used in the It is used in the leather industry. It is prepared by the reaction of sulfuric acid and copper. It is also obtained as a by-product from copper refineries.용도
Copper(II) sulfate may be employed for the following studies:- As a catalyst for the acetylation of alcohols and phenols under solvent-free conditions.
- To compose the electrolyte for the electrodeposition of Cu-Zn-Sn precursors, required for the preparation of Cu2ZnSnS4 (CZTS) thin films.
- As a Lewis acid catalyst for the dehydration of alcohols.5
정의
A compound prepared as the hydrate by the action of dilute sulfuric acid on copper( II) oxide or copper(II) carbonate. On crystallization, blue triclinic crystals of the pentahydrate (blue vitriol, CuSO4.5H2O) are formed. Industrially copper(II) sulfate is prepared by passing air through a hot mixture of dilute sulfuric acid and scrap copper. The solution formed is recycled until the concentration of the copper(II) sulfate is sufficient. Copper(II) sulfate is readily soluble in water. The monohydrate (CuSO4.H2O) is formed at 100°C and the anhydrous salt at 250°C. Anhydrous copper( II) sulfate is white; it is extremely hygroscopic and turns blue on absorption of water. It decomposes on heating to give copper(II) oxide and sulfur(VI) oxide.Copper(II) sulfate is used as a wood preservative, a fungicide (in Bordeaux mixture), and in the dyeing and electroplating industries.
일반 설명
A white or off-white solid. Melting point 200°C with decomposition. Non-combustible.공기와 물의 반응
Soluble in water.반응 프로필
Anhydrous Cupric sulfate serves as a weak oxidizing agent. Causes hydroxylamine to ignite. Gains water readily. The hydrated salt is vigorously reduced by hydroxylamine [Mellor 8:292(1946-1947)]. Both forms are incompatible with finely powdered metals. Both are incompatible with magnesium, corrode steel and iron, may react with alkalis, phosphates, acetylene gas, hydrazine, or nitromethane, and may react with beta-naphthol, propylene glycol, sulphathiazole and triethanolamine if the pH exceeds 7 . Both act as acidic salts, corrode metals and irritate tissues.위험도
Toxic; highly irritant.건강위험
INGESTION: copper sulfate may induce severe gastroenteric distress (vomiting, gastroenteric pain, and local corrosion and hemorrhages), prostration, anuria, hematuria, anemia, increase in white blood cells, icterus, coma, respiratory difficulties, and circulatory failure.농업용
Fungicide, Algaecide, Bactericide, Herbicide, Molluscicide: Copper sulfate is a fungicide used to control bacterial and fungal diseases of fruit, vegetable, nut, and field crops. These diseases include mildew, leaf spots, blights, and apple scab. It is used as a protective fungicide (Bordeaux mixture) for leaf application and seed treatment. It is also used as an algaecide and herbicide, and to kill slugs and snails in irrigation and municipal water treatment systems. It has been used to control Dutch elm disease. It is available as a dust, wettable powder, or liquid concentrate. Used as a fungicide and algaecide, in veterinary medicine and others. Copper sulfate is also used todetect and to remove trace amounts of water from alcohols and organic compounds.공업 용도
Copper sulfate (CuSO4·5H2O) is widely used as an activator for sphalerite, pyrite, pyrrhotite and other sulfides during processing of base metal ores. During flotation of some silicate minerals, copper sulfate is used as depressant, e.g. zirconium.In manufacturing copper sulfate, sulfuric acid and scrap copper metal are used. The process is based on the oxidation of metal and dissolution with H2SO4 according to the following reaction:
4Cu + O2 = 2Cu2O Cu2O + H2SO4 = CuSO4 + H2O 2Cu2SO4 + 2H2SO4 + O2 = 4CuSO4 + 2H2O Usually, in mineral processing applications, copper sulfate is delivered in crystal form.
상품명
AGRITOX®; BASICOP®; BCS COPPER FUNGICIDE®; BSC FLOWABLE®[C]; COPSIN®; CP BASIC SULFATE®; CUPROFIX®; FUNGI-SPERSE II[C]; SULTRACOB®; TNCS® 53; TRIANGLE®Safety Profile
A human poison by ingestion. An experimental poison by ingestion, subcutaneous, parenteral, intravenous, and intraperitoneal routes. Human systemic effects by ingestion: gastritis, Qarrhea, nausea or vomiting, damage to kidney tubules, and hemolysis. Questionable carcinogen with experimental tumorigenic data. An experimental teratogen. Other experimental reproductive effects. Mutation data reported. Reacts violently with hydroxylamine, magnesium. See also COPPER COMPOUNDS and SULFATES. When heated to decomposition it emits toxic fumes of SOx잠재적 노출
Copper sulfate is used as intermediate and wood preservative; also used in production of copper compounds; to detect and to remove trace amounts of water from alcohols and organic compounds; as a fungicide and algicide; in veterinary medicine and others.저장
Workers should keep copper sulfate stored in a cool, dry area with suffi cient ventilation. It should be kept away from alkalis, magnesium, ammonia, acetylene, and sodium hypobromite.운송 방법
UN3288 Toxic solids, inorganic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required. UN3077 Environmentally hazardous substances, solid, n.o.s., Hazard class: 9; Labels: 9-Miscellaneous hazardous material, Technical Name Required.Purification Methods
After adding 0.02g of KOH to a litre of nearly saturated aqueous solution of the sulfate, it is left for two weeks, then the precipitate is filtered on to a fibreglass filter with pore diameter of 5-15 microns. The filtrate is heated to 90o and allowed to evaporate until some CuSO4.5H2O crystallises out. The solution is then filtered hot and cooled rapidly to give crystals which are freed from mother liquor by filtering under suction [Geballe & Giauque J Am Chem Soc 74 3513 1952]. Alternatively crystallise the sulfate from water (0.6mL/g) between 100o and 0o. The pentahydrate is slowly efflorescent, losing 2H2O at 30o, two more H2O are lost at 110o and a white anhydrous powder (dessicant) is obtained on heating above 250o.비 호환성
Aqueous solution is an acid. May form explosive materials on contact with acetylene and nitromethane. Incompatible with strong bases; hydroxylamine, magnesium; zirconium, sodium hypobromite, hydrazine.폐기물 처리
Copper-containing soluble wastes can be concentrated through the use of ion exchange, reverse osmosis, or evaporators to the point where copper can be electrolytically removed and sent to a reclaiming firm. If recovery is not feasible, the copper can be precipitated through the use of caustics and the sludge deposited in a chemical waste landfill Add soda ash to waste CuSO4 solution; let stand 24 hours. Decant and neutralize solution before flushing to sewer. Landfill sludge.주의 사항
During handling and use of copper sulfate, students and occupational workers should wear safety glasses and should not breathe the material in powder form. Copper sulfate is an environmental pollutant and must be carefully incorporated when used in its varied applications. Workers should wear protective clothing, goggles, impermeable gloves, and rubber boots to avoid skin contact황산동(II) 준비 용품 및 원자재
원자재
준비 용품
polyamide conductive fiber
싸이오사이안산동(I)
5-FLUORO-2-HYDROXY-3-METHYLBENZALDEHYDE
Direct Blend Blue
2-(2-CHLOROETHOXY)-BENZENESULFONAMIDE
플로로페놀
2-CYANO-5-METHYLBENZENESULFONYL CHLORIDE
4-Chlorobenzenesulfonic acid
2-Bromo-5-nitrothiazole
Reactive Blue BRF
zinc plating additive DPE-1
프탈이드
ETHYL 2-CHLORO-4-METHYL-1,3-THIAZOLE-5-CARBOXYLATE
용제 비올렛 8
청색 103 반응염료
4-NITROPHENYL PHENYL ETHER
Sulphur Brown 4
1,2:3,4-Di-O-isopropylidene-D-galactopyranose
4-FLUORO-2-NITROBENZONITRILE
카퍼글루코네이트
자색 005 반응염료
3-NITRO-PYRIDINE-2-CARBOXYLIC ACID
Disperse Red 86
Rubber peptizer
3-PHENOXYTOLUENE
trisodium [5-acetamido-4-hydroxy-3-[[2-hydroxy-5-[[2-(sulphooxy)ethyl]sulphonyl]phenyl]azo]naphthalene-2,7-disulphonato(5-)]cuprate(3-)
trisodium [2-[(4,6-dichloro-1,3,5-triazin-2-yl)amino]-5-hydroxy-6-[(2-hydroxy-5-sulphophenyl)azo]naphthalene-1,7-disulphonato(5-)]cuprate(3-)
5-chloro-2-methylbenzenediazonium
구리 수산화물
Reactive Black 31
CuO-Al inorganic adhesive
1-METHYLTHYMINE
구리 파이로인산염
산성청색62호
Reactive Violet 2
5-CHLORO-2-NITROANISOLE
2-에틸헥소산 구리
6-BROMOPHTHALAZIN-1(2H)-ONE
tetrasodium [mu-[[7,7'-(carbonyldiimino)bis[4-hydroxy-3-[(2-hydroxy-5-sulphophenyl)azo]naphthalene-2-sulphonato]](8-)]]dicuprate(4-)
tetraammonium [mu-[[5,5'-[carbonylbis[imino(2-hydroxy-5-sulpho-p-phenylene)azo]]bis[6-hydroxynaphthalene-2-sulphonato]](8-)]]dicuprate(4-)
황산동(II) 공급 업체
글로벌( 640)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12456 | 58 |
| Shandong Juchuang Chemical Co., LTD | +86-18885615001 +86-18885615001 |
admin@juchuangchem.com | China | 387 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 |
fandachem@gmail.com | China | 9348 | 55 |
| Zjartschem | +86-571-8723 8903 jocelynpan@zjarts.com |
jocelynpan@zjarts.com | CHINA | 993 | 58 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 |
alice@crovellbio.com | China | 8823 | 58 |
| Xiamen AmoyChem Co., Ltd | +86-592-6051114 +8618959220845 |
sales@amoychem.com | China | 6387 | 58 |
| HubeiwidelychemicaltechnologyCo.,Ltd | 18627774460 |
faith@widelychemical.com | CHINA | 742 | 58 |