오메프라졸
|
|
오메프라졸 속성
- 녹는점
- 156°C
- 끓는 점
- 600.0±60.0 °C(Predicted)
- 밀도
- 1.332 g/cm3
- 인화점
- 9℃
- 저장 조건
- 2-8°C
- 용해도
- H2O: 0.5 mg/mL
- 물리적 상태
- 고체
- 물리적 상태
- 단단한 모양
- 산도 계수 (pKa)
- pKa 4.14/8.9(H2O,t =25,I=0.025) (Uncertain)
- 색상
- 하얀색
- 수용성
- 물(0.5mg/ml), DMSO(25mg/ml) 및 에탄올(4.5mg/ml)에 용해됩니다.
- Merck
- 14,6845
- BCS Class
- 2
- 안정성
- 안정적이지만 흡습성이 있고 감광성이 있습니다. 강한 산화제와 호환되지 않습니다. 어둠 속에 보관하십시오.
- InChIKey
- SUBDBMMJDZJVOS-UHFFFAOYSA-N
- CAS 데이터베이스
- 73590-58-6(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xi,T,F | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 36/37/38-39/23/24/25-23/24/25-11 | ||
| 안전지침서 | 26-36-37/39-45-36/37-16-7 | ||
| 유엔번호(UN No.) | UN1230 - class 3 - PG 2 - Methanol, solution | ||
| WGK 독일 | 2 | ||
| RTECS 번호 | DD9087000 | ||
| HS 번호 | 29333990 | ||
| 유해 물질 데이터 | 73590-58-6(Hazardous Substances Data) | ||
| 독성 | LD50 in mice, rats (g/kg): 0.08, >0.05 i.v.; >4, >4 orally (Ekman) |
오메프라졸 C화학적 특성, 용도, 생산
개요
오메프라졸(Omeprazole)은 소화성 궤양 질환, 소화불량, 위식도 역류질환, 역류성 인후두염, 졸린거-엘리슨증후군(Zollinger-Ellison syndrome)에 사용하는 약물로, 양성자 펌프 저해제(proton pump inhibitor)다. WHO(세계보건기구)가 선정한 필수 약물에 속한다.개요
오메프라졸은 위 식도 역류 질환, 소화성 궤양, 부식성 식도염 및 졸 린저 엘리슨 증후군의 치료에 사용될 수 있습니다.작용 기전
오메프라졸을 포함하는 양성자 펌프 저해제는 모든 제제가 비슷한 구조다. 양성자 펌프 저해제는 위의 벽세포에 존재하는 양성자 펌프에 비가역적으로 결합함으로써 위산이 분비되지 못하게한다.개요
Omeprazole is a potent gastric antisecretory agent with selective inhibitory effect on the H+,K+-ATPase proton pump. It is highly effective in the treatment of duodenal ulcer and Zollinger-Ellison syndrome, and is reportedly superior to ranitidine in the management of reflux esophagitis.화학적 성질
White Crystalline Solid용도
Omeprazole is a proton pump inhibitor used to treat diseases like gastroesophageal reflux disease (GERD), used for gastric and duodenal ulcers, reflux or erosive esophagitis, and Zollinger-Ellison syndrome. It is also effective for gastric and duodenal ulcers that are ineffective with H2 receptor antagonists. Injections of Omeprazole can also be used for: 1 gastrointestinal bleeding, such as peptic and anastomic ulcer bleeding, and the prevention of severe diseases (such as cerebral hemorrhage, severe trauma, etc.) and gastric surgery caused by upper intestinal bleeding; 2 acute gastric mucosal damage complicated by stress or nonsteroidal anti-inflammatory drugs; 3 general anesthesia, post-surgery, or coma patients, to prevent acid reflux and aspiration pneumonia; 4 Combined with amoxicillin and clarithromycin, or with metronidazole and clarithromycin, it can effectively kill Helicobacter pylori (Hp).정의
ChEBI: Omeprazole is a member of the class of benzimidazoles that is 1H-benzimidazole which is substituted by a [4-methoxy-3,5-dimethylpyridin-2-yl)methyl]sulfinyl group at position 2 and a methoxy group at position 5.제조 방법
The antiulcer agent omeprazole is produced from 2,3,5-trimethylpyridine N-oxide.Synthesis and Structure of Omeprazole
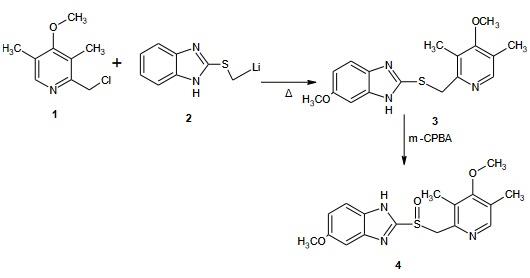
Steps: 2-(Lithium methyl sulphinyl)-5-methoxy-1H benzimidazole 20g was reacted with 2-chloro-3,5-dimethyl-4-methoxy pyridine 21 g to form sulphide intermediate and then converted to Omeprazole when treated with m-CPBA which used as anoxidizingagents. The acetamide-sulfide compounds modification are oxidised to form the amide sulfinyl compound and gives the sulfinyl carboxylate or salts upon alkaline hydrolysis.On further decarboxylation leads to the target molecules. The residual, unreacted salt, inorganic by-products and other minor by-products can be easily purified by a simple washing from omeprazole or lansoprazole. The amide compounds containing crystalline solids as opposed to the sulphide and sulfoxides of the reported procedures.
DOI: http://dx.doi.org/10.20902/IJPTR.2019.120307
주요 응용
Omeprazole is a benzimidazole with selective and irreversible proton pump inhibition activity. Omeprazole forms a stable disulfide bond with the sulfhydryl group of the hydrogen-potassium (H+ - K+) ATPase found on the secretory surface of parietal cells, thereby inhibiting the final transport of hydrogen ions (via exchange with potassium ions) into the gastric lumen and suppressing gastric acid secretion. This agent exhibits no anticholinergic activities and does not antagonize histamine H2 receptors. Omeprazole Pellets are used in the treatment of Gastroesophageal reflux disease (GERD): A condition in which backward flow of acid from the stomach causes heartburn and injury of the food pipe (esophagus).World Health Organization (WHO)
Omeprazole was introduced in the 1980s. It belongs to a group of agents that have an inhibitory effect on the secretion of hydrochloric acid in the stomach (gastric acid proton pump inhibitors) and is used in the treatment of upper gastrointestinal tract disorders. The Committee for Proprietary Medicinal Products of the European Commission has concluded that a causal association between the reactions reported in Germany and the use of omeprazole had not been established. Nevertheless oral administration should be preferred. (Reference: (CPMPPO) Pharmacovigilance Opinion, No.16 , , 25 July 1994)일반 설명
Omeprazole, 5-methoxy-2-(((4-methoxy-3, 5-dimethyl-2-pyridinyl)methyl) sulfinyl)-1Hbenzimidazole(Losec), is a white to off-white crystallinepowder with very slight solubility in water. Omeprazole isan amphoteric compound (pyridine N, pKa 4.06; benzimidazoleN-H, pKa 0.79), and consistent with the proposedmechanism of action of the substituted benzimidazoles, isacid labile. Hence, the omeprazole product is formulatedas delayed-release capsules containing enteric-coatedgranules.The absolute bioavailability of orally administeredorneprazole is 30% to 40% related to substantial first-passbiotransformation. The drug has a plasmahalf-life of about 1 hour. Most (77%) of an oral dose ofomeprazole is excreted in the urine as metabolites with insignificantantisecretory activity. The primary metabolitesof omeprazole are 5-hydroxyomeprazole (CYP2C19) andomeprazole sulfone (CYP3A4). The antisecretory actions ofomeprazole persist for 24 to 72 hours, long after the drughas disappeared from plasma, which is consistent with itssuggested mechanism of action involving irreversible inhibitionof the proton pump.
Omeprazole is approved for the treatment of heartburn,GERD, duodenal ulcer, erosive esophagitis, gastric ulcer,and pathological hypersecretory conditions.
생물학적 활성
H + ,K + -ATPase inhibitor (IC 50 = 5.8 μ M) that displays antisecretory and antiulcer activity. Inhibits gastric acid secretion (IC 50 = 0.16 μ M for histamine-induced acid formation) and reduces gastric lesion formation induced by a variety of ulcerative stimuli. Antibacteral against Helicobacter pylori in vitro . Also inhibits CYP2C19, CYP2C9 and CYP3A (K i values are 3.1, 40.1 and 84.4 μ M respectively) and blocks swelling-dependent chloride channels (ICIswell).Clinical Use
Omeprazole is a proton-pump inhibitor used in the management and treatment of several conditions, including uncomplicated heartburn, peptic ulcer disease, gastrointestinal reflux disease, Zollinger-Ellison syndrome, multiple endocrine adenomas, systemic mastocytosis, erosive esophagitis, gastric ulcers, and helicobacter pylori infection.신진 대사 경로
When male humans are given 14C-omeprazole orally, an average of 79% of the dose is recovered in the urine in 96 h. Omeprazole is completely metabolized and at least six metabolites are identified. Two major metabolites are hydroxyomeprazole and omeprazole acid.Mode of action
Omeprazole is a proton pump inhibitor which can specifically act on gastric parietal cell proton pump sites and transform into the active form of sulfonamide, then irreversibly binds to the proton pumps through disulfide bonds, generating a sulfonamide and proton pump compound (H + -K + -ATP), thereby inhibiting the enzymatic activity, preventing the H+ in parietal cells from being transported to the stomach cavity. It has a strong and persistent inhibitory role on gastric acid secretion caused by basal gastric acid and pentapeptide gastric acid secretions, greatly reducing gastric acid within the gastric juice. Rapid, reversible, and no H2 antagonist-induced psychiatric side effects.오메프라졸 준비 용품 및 원자재
원자재
에틸 크산토겐산 칼륨
1H-벤즈아미다졸
3-클로로페록시벤조산
클로로포름
3,5-Dimethyl-4-methoxy-2-pyridinemethanol
2-MERCAPTO-5-METHOXYBENZIMIDAZOLE
O-페닐렌다이아민
디메틸설페이트
염화 티오닐
Benzylamine hydrochloride
무수프탈산
과황산암모늄
2-Mercapto-5-methylbenzimidazole
과산화 수소
준비 용품
오메프라졸 공급 업체
글로벌( 773)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Lingding Biotechnology Co., Ltd. | +86-18031140164 +86-19933155420 |
erin@hbldbiotech.com | China | 879 | 58 |
| Shenzhen Excellent Biomedical Technology Co.,Ltd. | +86-0755-26050679 +86-15915472436 |
sale@ex-biotech.cn | China | 1031 | 58 |
| Sinoway Industrial co., ltd. | 0592-5800732; +8613806035118 |
xie@china-sinoway.com | China | 992 | 58 |
| Anhui Yiao New Material Technology Co., Ltd | +86-199-55145978 +8619955145978 |
sales8@anhuiyiao.com | China | 253 | 58 |
| Henan Tengmao Chemical Technology Co. LTD | +8615238638457 |
salesvip2@hntmhg.com | China | 415 | 58 |
| Wuhan Haorong Biotechnology Co.,ltd | +8618565342920 |
sales@chembj.net | China | 269 | 58 |
| Hebei Anlijie Biotechnology Co., Ltd | +8619031013551 |
ably@aljbio.com | China | 177 | 58 |
| Sigma Audley | +86-18336680971 +86-18126314766 |
nova@sh-teruiop.com | China | 524 | 58 |
| Shanghai Affida new material science and technology center | +undefined15081010295 |
2691956269@qq.com | China | 359 | 58 |
| Hunan aslsen technology co.,ltd | +8617397237530 |
aslsa@aslsen.com | China | 124 | 58 |