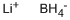리튬 보로수소화물
|
|
리튬 보로수소화물 속성
- 녹는점
- 280 °C
- 끓는 점
- 66°C/760mmHg
- 밀도
- 0.896 g/mL at 25 °C
- 인화점
- −1 °F
- 저장 조건
- water-free area
- 용해도
- 에테르, THF 및 지방족 아민에 용해되며 에테르, 테트라히드로푸란, 지방족 아민 및 에탄올에 용해됩니다.
- 물리적 상태
- 가루
- Specific Gravity
- 0.66
- 색상
- 하얀색
- 폭발한계
- 4.00-75.60%(V)
- 수용성
- pH 7 이상의 가용성 H2O, 에테르, 테트라히드로푸란, 지방족 아민 [MER06]
- 감도
- Air & Moisture Sensitive
- Merck
- 14,5525
- CAS 데이터베이스
- 16949-15-8(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | F,T,C,Xn,F+ | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 14/15-23/24/25-34-20/21/22-11-40-36/37/38-19-67-66-22-12 | ||
| 안전지침서 | 26-36/37/39-43-45-36/37-16 | ||
| 유엔번호(UN No.) | UN 3399 4.3/PG 1 | ||
| WGK 독일 | 2 | ||
| RTECS 번호 | ED2725000 | ||
| F 고인화성물질 | 10-21 | ||
| TSCA | Yes | ||
| HS 번호 | 2850 00 20 | ||
| 위험 등급 | 4.3 | ||
| 포장분류 | I | ||
| 기존화학 물질 | KE-22598 |
리튬 보로수소화물 C화학적 특성, 용도, 생산
화학적 성질
WHITE POWDER물리적 성질
White orthorhombic crystals; density 0.67 g/cm3; decomposes in moist air; melts at 268°C; decomposes at 380°C; reacts with water; dissolves in ether, tetrahydrofuran, and diethylamine; solubility in ether, 25g/L at 25°C.용도
Strong reducing agent. Used to reduce compounds containing ketones, aldehydes, ester carbonyls, acid chlorides, lactones, and epoxidesLithium borohydride is a versatile reducing agent for aldehydes, ketones, nitriles, primary amides, acid chlorides, lactones, epoxides, and esters. It is involved in the preparation of intercalation compounds and nanocomposites. It is used as a precursor for other borohydrides and acts as a catalyst in hydroboration reactions. It is known to be good reversible storage compound for anhydrous ammonia and hydrogen.일반 설명
A white to grayish crystalline powder.공기와 물의 반응
Likely to ignite when moistened with water [Lab. Gov. Chemist 1965].반응 프로필
Lithium borohydride is a strong reducing agent. Is easily ignited and burns vigorously once ignited. Reacts on contact with water or acids to form hydrogen gas and corrosive products. Reaction with limited amounts of water or moisture may cause ignition after a delay [Gaylord, 1965, p. 22].건강위험
Inhalation or contact with vapors, substance or decomposition products may cause severe injury or death. May produce corrosive solutions on contact with water. Fire will produce irritating, corrosive and/or toxic gases. Runoff from fire control may cause pollution.화재위험
Produce flammable gases on contact with water. May ignite on contact with water or moist air. Some react vigorously or explosively on contact with water. May be ignited by heat, sparks or flames. May re-ignite after fire is extinguished. Some are transported in highly flammable liquids. Runoff may create fire or explosion hazard.Safety Profile
Poison by ingestion, inhalation, and skin contact. Flammable; can liberate H2. Incompatible with H20 as moisture on fibers of cellulose or as liquid. See also LITHIUM, BORON COMPOUNDS, and HYDRIDES.Purification Methods
It is crystallised from Et2O, and pumped free of ether at 90-100o during 2hours [Schaeffer et al. J Am Chem Soc 78 729 1956]. Store it dry as it decomposes slowly in moist air. [Becher in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I p 775 1963.]리튬 보로수소화물 준비 용품 및 원자재
원자재
준비 용품
BENZYL 4-(BROMOMETHYL)TETRAHYDRO-1(2H)-PYRIDINECARBOXYLATE
3,5-Dimethoxybenzylamine
Methyl 3-formylbenzoate
Thiophene-2-ethylamine
1-AMINO-CYCLOPROPANEMETHANOL
1H-Azepine-3-carboxylicacid,hexahydro-,methylester(9CI)
2,4-DIMETHOXYPHENETHYLAMINE
(S)-4-BOC-MORPHOLINE-3-CARBOXYLIC ACID
5-(AMINOMETHYL)-1-METHYLPYRROLIDIN-2-ONE
리튬 보로수소화물 관련 검색:
산화 리튬 나트륨 사이아노브로민하이드라이드(나트륨 시아노브롬하이드리드) 칼륨보로하이드라이드 염화 리튬 나트륨 붕소수화물 수소화리튬 리튬 테트라플루오로붕산염 붕소 리튬 보로수소화물 리튬 리튬트리에틸보로하이드라이드용액 테트라하이드로푸란
LITHIUM OXALATE
Sodium triacetoxyborohydride
LITHIUM TETRAPHENYLBORATE TRIS(1,2-DIMETHOXYETHANE)
Lithium bis(oxalate)borate
LITHIUM TETRAKIS(PENTAFLUOROPHENYL)BORATE-ETHYL ETHER COMPLEX
LITHIUM 9-BBN HYDRIDE