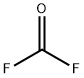
플루오린화 카보닐 C화학적 특성, 용도, 생산
화학적 성질
Carbonyl fluoride is colorless or light yellow,
hygroscopic, compressed liquefied gas. Pungent, highly
irritating and suffocating odor.
물리적 성질
Colorless gas; pungent odor; hygroscopic; unstable; liquid density 1.139 g/mL (at -114°C); liquefies at -83.1°C; solidifies at -114°C; decomposes in water.
용도
Organic synthesis.
제조 방법
Carbonyl fluoride is prepared by the reaction of carbon monoxide with fluorine gas or silver fluoride:
CO + F
2 → COF
2 Also, it may be produced by the action of carbon monoxide with bromine trifluoride, BrF
3.
일반 설명
A colorless gas with a pungent odor. Very toxic by inhalation. Prolonged exposure of the containers to fire or heat may result in violent rupturing and rocketing.
공기와 물의 반응
Reacts with water or steam to produces corrosive and toxic hydrofluoric acid fumes.
반응 프로필
CARBONYL FLUORIDE is an acid fluoride. Incompatible with water, with bases (including amines), with strong oxidizing agents, with alcohols. Reacts violently with hexafluoroisopropylideneaminolithium. High temperature causes decomposition to toxic carbon monoxide gas and fluorine. May react vigorously or explosively if mixed with diisopropyl ether or other ethers in the presence of trace amounts of metal salts [J. Haz. Mat., 1981, 4, 291].
위험도
Toxic by inhalation, strong irritant to skin.
Lower respiratory tract irritant. Bone damage.
건강위험
Irritates lungs, causing delayed pulmonary edema. Slight gassing produces dryness or burning sensation in the throat, numbness, pain in the chest, bronchitis, and shortness of breath.
화재위험
Special Hazards of Combustion Products: Toxic gas is generated when heated.
Safety Profile
A poison. Moderately
toxic by inhalation. A powerful irritant.
Hydrolyzes instantly to form HF on contact
with moisture. See also CARBONYLS,
HYDROFLUORIC ACID, and
FLUORINE. Incompatible with
hexafluoroisoprop ylideneamino-lithium.
When heated to decomposition it emits
toxic fumes of CO and F-. See CARBON
MONOXIDE for fire and explosion hazard.
잠재적 노출
Carbonyl fluoride is a carboxy halide.
The major source of exposure to COF2 results from the
thermal decomposition of fluoro carbon plastics, such as
PTFE in air. Carbonyl fluoride is used for synthesizing
fluoroalkanes, difluoroisocyanates, and fluorinated alkyl
isocyanates. It may have been used as a military poison
gas.
운송 방법
UN2417 Carbonyl fluoride, Hazard class: 2.3;
Labels: 2.3-Poisonous gas, 8-Corrosive material, Inhalation
Hazard Zone B. Cylinders must be transported in a secure
upright position, in a well-ventilated truck. Protect cylinder
and labels from physical damage. The owner of the compressed gas cylinder is the only entity allowed by federal
law (49CFR) to transport and refill them. It is a violation
of transportation regulations to refill compressed gas cylinders without the express written permission of the owner.
비 호환성
Reacts with water to form toxic and corrosive HF gas. HF gas is highly reactive and forms explosive hydrogen gas on contact with metals. Do not use cast
iron or malleable fittings with carbonyl fluoride. Carbonyl
fluoride decomposes on heating above 450C producing
toxic gases, including HF. Not compatible with
hexafluoroisopropylidene-amino lithium; reaction may be
dangerous.
폐기물 처리
Return refillable compressed
gas cylinders to supplier.
플루오린화 카보닐 준비 용품 및 원자재
원자재
Hexanal, 3,3,4,4,5,5,6,6,6-nonafluoro-
Hexanoic acid, 3,3,4,4,5,5,6,6,6-nonafluoro-
1H,1H,2H,2H-Perfluorohexan-1-ol
PERFLUOROPENTANOIC ACID
디플루오로디브로모메탄
헥사플루오린화 우라늄(헥사플루오르화 우라늄)
CARBONYL CHLORIDE FLUORIDE
준비 용품