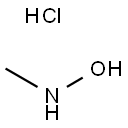
N-메틸히드록실아민 수화염화물
|
|
N-메틸히드록실아민 수화염화물 속성
- 녹는점
- 86-88 °C(lit.)
- 밀도
- 1.36 g/cm3
- 저장 조건
- Inert atmosphere,Room Temperature
- 용해도
- H2O: 0.1 g/mL, 투명, 매우 희미한 노란색
- 물리적 상태
- 고체
- 물리적 상태
- 단단한 모양
- 색상
- 하얀색
- 수용성
- 녹는
- 감도
- Hygroscopic
- BRN
- 3541409
- CAS 데이터베이스
- 4229-44-1(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 36/37/38 | ||
| 안전지침서 | 26-36-24/25 | ||
| 유엔번호(UN No.) | 1759 | ||
| WGK 독일 | 3 | ||
| F 고인화성물질 | 3-10 | ||
| TSCA | Yes | ||
| 위험 등급 | 8 | ||
| 포장분류 | III | ||
| HS 번호 | 29280090 | ||
| 기존화학 물질 | KE-20610 |
N-메틸히드록실아민 수화염화물 C화학적 특성, 용도, 생산
화학적 성질
The substance appears as white to almost white crystals or crystalline powder. It exhibits solubility in water.용도
N-Methylhydroxylamine hydrochloride is used as an inorganic catalyst used in the transamidation of primary amides with amines. It is employed in constructing hydroxamates, important functional groups for the complexation of iron.참고 문헌
[1] A. Gibson and S. Mirzazadeh. N-methylhydroxylamine inhibits and M&B 22948 potentiates relaxations of the mouse anococcygeus to non-adrenergic, non-cholinergic field stimulation and to nitrovasodilator drugs.British Journal of Pharmacology.1989, 96 637-644. DOI:10.1111/j.1476-5381.1989.tb11863.x[2] J. R. Ochoa Gomez. Electrosynthesis of N-methylhydroxylamine.Journal of Applied Electrochemistry.1991, 21 331-334. DOI:10.1007/BF01020218
[3] Reacts with aldehydes and ketones to give nitrones, which undergo 1,3-dipolar addition reactions with alkenes. Cycloaddition to trimethylvinylsilane has been used in a 2-carbon extension of aldehydes to ɑ?-unsaturated aldehydes: J. Org. Chem., 49, 3421 (1984). DOI:10.1021/jo00192a047
[4] Intramolecular cycloaddition has been used as a route to bicyclic systems, e.g. from citronellal: Org. Synth. Coll., 6, 670 (1988).
[5] Reviews: 1,3-Dipolar cycloadditions of nitrones: Synthesis, 205 (1975). The [3+2] nitrone-olefin cycloaddition reaction: Org. React., 36, 1 (1988). Synthetic applications of nitrones: Org. Prep. Proced. Int., 17, 25 (1985).
[6] For conversion to, and reactions of, the N,O-bis(TMS) derivative, see: J. Chem. Soc., Perkin 1, 1823 (1989).
[7] GAN Y, WENKUI Z, HUANG H, et al. Industrial Synthesis of N-Methylhydroxylamine Hydrochloride by Electrochemical Reduction of Nitromethane[J]. Chinese Journal of Chemical Engineering, 2006, 14: 649-653. DOI:10.1016/S1004-9541(06)60129-8.
N-메틸히드록실아민 수화염화물 준비 용품 및 원자재
원자재
준비 용품
N-메틸히드록실아민 수화염화물 공급 업체
글로벌( 252)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| Accela ChemBio Inc. | (+1)-858-699-3322 |
info@accelachem.com | United States | 19965 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-61398051 +8613650506873 |
sales@chemdad.com | China | 39916 | 58 |
| Alchem Pharmtech,Inc. | 8485655694 |
sales@alchempharmtech.com | United States | 63711 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 |
sales@conier.com | China | 47465 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-29-87569266 15319487004 |
1015@dideu.com | China | 2263 | 58 |
| Antai Fine Chemical Technology Co.,Limited | 18503026267 |
info@antaichem.com | CHINA | 9641 | 58 |
| Neostar United (Changzhou) Industrial Co., Ltd. | +86-519-519-85557386 |
marketing1@neostarunited.com | China | 8349 | 58 |
| Zhengzhou Alfa Chemical Co.,Ltd | +8618530059196 |
sale04@alfachem.cn | China | 12468 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | 0551-65418671 |
sales@tnjchem.com | China | 34572 | 58 |