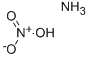Ammonium nitrate suppliers
Ammonium nitrate
- CAS:
- 6484-52-2
- MF:
- H4N2O3
- MW:
- 80.04
Properties
- Melting point:
- 169 °C(lit.)
- Boiling point:
- 210 °C(lit.)
- Density
- 1.72
- Flash point:
- 210°C
- storage temp.
- Store at RT.
- solubility
- H2O: 1 M at 20 °C, clear, colorless
- form
- Solid
- Specific Gravity
- 1.725
- color
- White
- PH
- 4.5-6.0 (25℃, 50mg/mL in H2O)
- Water Solubility
- 190 g/100 mL (20 ºC)
- Merck
- 14,534
Safety Information
- Symbol(GHS)


GHS03,GHS07
- Signal word
- Warning
- Hazard statements
- H272-H319
- Precautionary statements
- P210-P220-P264-P280-P305+P351+P338-P337+P313
- Hazard Codes
- O,Xi
- Risk Statements
- 8-36/37/38-9
- Safety Statements
- 17-26-36-37/39-41-16-15
- RIDADR
- UN 1942 5.1/PG 3
- WGK Germany
- 1
- RTECS
- BR9050000
- F
- 3-10
- TSCA
- Yes
- HazardClass
- 5.1
- PackingGroup
- III
- HS Code
- 31023090
- Toxicity
- LD50 orally in Rabbit: 2462 mg/kg
Use
Ammonium nitrate (NH4NO3, also known as “Norway saltpeter”) is mainly used as a fertilizer. It is also known as the chemical that was mixed with diesel fuel to create the explosion that demolished the Murrah Federal Building in Oklahoma City in 1995. It provides a source of nitrogen to plants to increase growth and crop yields. Ammonium nitrate is a popular fertilizer since it provides half of the N in nitrate form and half in ammonium form. The nitrate form moves readily with soil water to the roots where it is immediately available for plant uptake. Ammonium nitrate is more popular than other nitrogen fertilizers for pasture and hay fertilization since it is less susceptible to volatilization or air losses when left on the soil. It is also valued by vegetable growers for its ability to provide an immediately available nitrate source of plant food.
According to relevant laws, regulations, policies and the provisions of this website, this website does not display the product information!
The source is "易制爆、炸药、化学武器等". If the information is inaccurate, please contact the platform to modify it.
contact information:info@chemicalbook.com 400-158-660