| | 873054-44-5 Basic information More.. |
| Product Name: | VX-770 | | Synonyms: | Ivacaftor, >=98%;3-QuinolinecarboxaMide, N-[2,4-bis(1,1-diMethylethyl)-5-hydroxyphenyl]-1,4-dihydro-4-oxo-;N-[2,4-Bis(tert-butyl)-5-hydroxyphenyl]-1,4-dihydro-4-oxo-3-quinolinecarboxamide;VX 770, Ivacaftor;Ivacaftor-D4;VX-770(Ivacattor);IVACAFTOR (VX-770);VX 770;N-(2,4-DITERT-BUTYL-5-HYDROXYPHENYL)-4-OXO-1H-QUINOLINE-3-CARBOXAMIDE | | CAS: | 873054-44-5 | | MF: | C24H28N2O3 | | MW: | 392.49 | | EINECS: | | | Mol File: | 873054-44-5.mol | 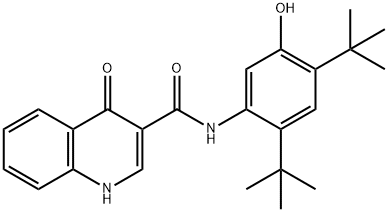 |
Use
In January 2012, the US FDA approved ivacaftor for the treatment of cystic
fibrosis (CF) in patients who have the G551D mutation of the CF transmembrane regulator (CFTR) and are at least 6 years old. Ivacaftor (also known as VX-770) is a CFTR potentiator that increases the open probability of CFTR, thus increasing chloride
secretion particularly in the 5% of CF patients with the G551D/F508 gating/
processing mutation. Ivacaftor was discovered by medicinal chemistry optimization of a lead scaffold identified through high-throughput screening of a 228,000 compound collection. In cultured bronchial epithelial cells from a CF patient with F508del, ivacaftor increased chloride secretion
(EC50=81 nM). Preparation of ivacaftor is accomplished via a multistep
synthesis oftwointermediates, 4-oxo-1,4-dihydroquinoline-3-carboxylic acid
and 5-amino-di-tert-butylphenyl methyl carbonate, which are coupled using
propane phosphonic acid anhydride (T3P) to afford the amide; deprotection of
the phenol then provides ivacaftor.
- VX-770
-

- US $1.10 / g
- 2022-05-24
- CAS:873054-44-5
- Min. Order: 1g
- Purity: 99.0% Min
- Supply Ability: 100 Tons
- Ivacaftor
-

- US $15.00-10.00 / KG
- 2021-07-13
- CAS:873054-44-5
- Min. Order: 1KG
- Purity: 99%+ HPLC
- Supply Ability: Monthly supply of 1 ton
- Ivacaftor
-

- US $15.00-10.00 / KG
- 2021-07-10
- CAS:873054-44-5
- Min. Order: 1KG
- Purity: 99%+ HPLC
- Supply Ability: Monthly supply of 1 ton
|
|
873054-44-5
Recommend Suppliers |
|