|
|
| | 4-(3'-Methylphenyl)amino-3-pyridinesulfonamide Basic information |
| | 4-(3'-Methylphenyl)amino-3-pyridinesulfonamide Chemical Properties |
| Melting point | 162-164?C | | Boiling point | 475.8±55.0 °C(Predicted) | | density | 1.357±0.06 g/cm3(Predicted) | | storage temp. | Keep in dark place,Inert atmosphere,Room temperature | | solubility | DMSO (Slightly), Methanol (Slightly) | | pka | 9.53±0.60(Predicted) | | color | White to Off-White | | BRN | 5438165 | | InChIKey | ZXPCUGWAKUIOOF-UHFFFAOYSA-N | | CAS DataBase Reference | 72811-73-5(CAS DataBase Reference) |
| Hazard Codes | Xi | | Risk Statements | 36 | | Safety Statements | 26 | | HS Code | 2935909550 |
| | 4-(3'-Methylphenyl)amino-3-pyridinesulfonamide Usage And Synthesis |
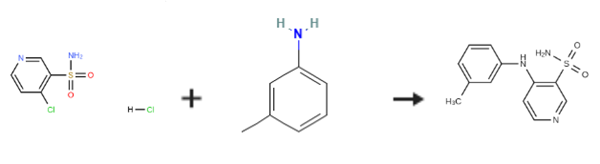
| Chemical Properties | White to Off-White Solid | | Uses | Torasemide intermediate. | | Synthesis | 4-(3'-Methylphenyl)amino-3-pyridinesulfonamide is prepared by the reaction of 4-Chloropyridine-3-sulfonamide hydrochloride and 3-Methylaniline. The specific synthesis steps are as follows:
EXAMPLE 1; Preparation of 4-(3-methylphenyl)aminopyridine-sulfonamide; 2L three-neck flask, equipped with a mechanical stirrer, thermometer and condenser, was charged with water (500 ml) and 4-chloro-3- pyridinesulfonamide hydrochloride (100g, 0.44 mol). To this suspension was added m-toluidine (49.2 ml, 0.46 mol) at room temperature. The reaction mixture was heated to 90°C for a minimum period of 3 h. The progress of the reaction was followed by HPLC. After completion, the mixture was cooled to room temperature. The pH of the reaction was then adjusted carefully to pH 7-8 with sat. NaHC03 (ca. 1.1 l). The product was precipitated out and isolated by vacuum filtration as beige solid (126.2 g wet weight). The product was then dissolved in MeOH (1.0 l) at room temperature and charged with Darco KB (25g). The solution was refluxed for 0.5 h and then filtered through a patch of celite to remove Darco KB, while still hot, and rinsed with hot MeOH (200 ML). The filtrate was then charged with water (1.2 l) and stirred for a minimum of 1 h at room temperature. The product, which had precipitated out, was isolated by vacuum filtration to obtain a solid 106.3 g (92percent wet weight =>99.8percent purity a/a). 1H NMR (d6-DMSO) ; 2.30 (s, 3H), 7.00-7. 15 (m. 5H), 7.32 (m, 1H), 7.75 (brs, 1.5H), 8.05 (brs, 0.5H), 8.25 (d, 1H), 8.68 (s, 1H).
 |
| | 4-(3'-Methylphenyl)amino-3-pyridinesulfonamide Preparation Products And Raw materials |
|