- Sodium Phosphate, Dibasic
-

- $6.00 / 1KG
-
2024-04-12
- CAS:7558-79-4
- Min. Order: 1KG
- Purity: More than 99%
- Supply Ability: 500KG/Month
- Sodium Phosphate, Dibasic
-
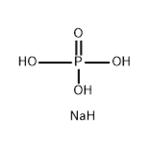
- $50.00 / 1KG
-
2023-12-23
- CAS:7558-79-4
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
|
| | Sodium Phosphate, Dibasic Basic information |
| Product Name: | Sodium Phosphate, Dibasic | | Synonyms: | Sodium phosphate dibasic Vetec(TM) reagent grade, 99%;di-Sodium hydrogen phosphate anhydrous for analysis EMSURE ACS,Reag. Ph Eur;Sodium phosphate dibasic anhydrous, free-flowing, Redi-Dri, ACS reagent, >=99%;SODIUM PHOSPHATE DIBASIC BIOXTRA;SODIUM PHOSPHATE DIBASIC SOLUTION 0.;SODIUM PHOSPHATE DIBASIC, BIOREAGENT;SODIUM PHOSPHATE DIBASIC, REAGENTPLU;SODIUM PHOSPHATE, DIBASIC MOLECULAR* | | CAS: | 7558-79-4 | | MF: | Na2HPO4 | | MW: | 141.96 | | EINECS: | 231-448-7 | | Product Categories: | metal phosphate compound;Chemistry;Inorganics;Food Additives;7558-79-4 | | Mol File: | 7558-79-4.mol | 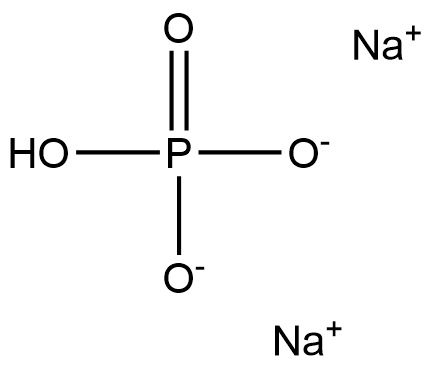 |
| | Sodium Phosphate, Dibasic Chemical Properties |
| Melting point | 243-245 °C | | density | 1.064 g/mL at 20 °C | | vapor density | 4.9 (vs air) | | vapor pressure | 0Pa at 20℃ | | FEMA | 2398 | DISODIUM PHOSPHATE | | storage temp. | Store at +5°C to +30°C. | | solubility | H2O: 0.5 M at 20 °C, clear, colorless | | form | powder | | color | White | | PH | 8.9-9.2 (25℃, 50g/L in water) | | pka | (1) 2.15, (2) 6.82, (3) 12.38 (at 25℃) | | Odor | odorless | | Water Solubility | >=10 g/100 mL at 20 ºC | | Sensitive | Hygroscopic | | λmax | λ: 260 nm Amax: 0.019
λ: 280 nm Amax: 0.016 | | Merck | 14,8659 | | Stability: | Stable. Incompatible with strong acids. Hygroscopic. | | InChIKey | BNIILDVGGAEEIG-UHFFFAOYSA-L | | LogP | -2.15 | | CAS DataBase Reference | 7558-79-4(CAS DataBase Reference) | | EPA Substance Registry System | Disodium phosphate (7558-79-4) |
| | Sodium Phosphate, Dibasic Usage And Synthesis |
| Description | Sodium phosphate dibasic is highly hygroscopic and water soluble. Useful in conjunction with Sodium Phosphate, Monobasic in the preparation of biological buffers. It is commonly used in biological assay buffers.
Sodium is required for nerve and muscle functioning. Sodium Phosphate Dibasic can be used as a dietary supplement and as a nutrient.
Sodium hydrogen phosphate is used in food and water treatment in conjunction with trisodium phosphate.In foods, it is used to adjust pH. In water treatment, It retards calcium scale formation. It is also found in some detergents and cleaning agents.
This product is known to be Used in many applications including the purification of antibodies. It is used as a saline laxative to treat constipation or to clean the bowel before a colonoscopy. | | Chemical Properties | Sodium hydrogen phosphate,NaH2P04·H20, is hygroscopic, transparent, water-soluble crystals. Used as a purgative, reagent,and buffer. | | Chemical Properties | The anhydrous compound is white crystalline powder; hygroscopic; density 1.70 g/cm3; converts to sodium pyrophosphate at 240°C; soluble in water; 874 SODIUM PHOSPHATE, DIBASIC insoluble in alcohol. The dihydrate is a crystalline powder or granular solid; density 2.066 g/cm3 at 15°C; loses water of crystallization at 92.5°C. The heptahydrate is a powdered or granular crystalline solid; density 1.679 g/cm3; loses five molecules of water at 48°C. The dodecahydrate is made up of translucent crystals or granules; density 1.524 g/cm3; readily loses five water molecules on exposure to air at ambient temperature; melts at 35°C when the salt contains all water of crystallization; loses all water at 100°C. All the hydrates are soluble in water and insoluble in alcohol. | | Chemical Properties | Disodium phosphate is anhydrous or contains two molecules of water of hydration | | Physical properties | The anhydrous compound is white crystalline powder; hygroscopic; density 1.70 g/cm3; converts to sodium pyrophosphate at 240°C; soluble in water;insoluble in alcohol.
The dihydrate is a crystalline powder or granular solid; density 2.066 g/cm3 at 15°C; loses water of crystallization at 92.5°C.
The heptahydrate is a powdered or granular crystalline solid; density 1.679 g/cm3; loses five molecules of water at 48°C.
The dodecahydrate is made up of translucent crystals or granules; density 1.524 g/cm3; readily loses five water molecules on exposure to air at ambient temperature; melts at 35°C when the salt contains all water of crystallization; loses all water at 100°C.
All the hydrates are soluble in water and insoluble in alcohol. | | Occurrence | Disodium phosphate is anhydrous or contains two molecules of water of hydration | | Uses | Sodium Phosphate Dibasic is used as an in conjunction with trisodium phosphate in foods and water treatment. In foods, it is used to adjust pH. In water treatment, It retards calcium scale formation. It is also found in some detergents and cleaning agents. Sodium Phosphate Dibasic is used as a saline laxative to treat constipation or to clean the bowel before a colonoscopy. | | Uses | It is used in pH buffer solutions. | | Uses | Sodium Phosphate, Dibasic is used as a laboratory reagent and a buffer in chemical analysis. Other uses are in manufacturing ceramics, detergents, and enamels; as a mordant in dyeing; for fireproofing paper and wood; for weighting and printing silk; in the treatment of boiler water; as a sequestrant in food; as a dietary supplement; in soldering enamels; and in fertilizers. It is used therapeutically as a cathartic. | | Preparation | Dibasic sodium phosphate is prepared by treating phosphoric acid with a slight excess of sodium carbonate. The solution is boiled to expel carbon dioxide. Upon cooling dodecahydrate crystallizes out. Heating dodecahydrate at 100°C forms the anhydrous salt.
H3PO4 + Na2CO3 → Na2HPO4 + CO2 + H2O
Dibasic sodium phosphate also is prepared by reacting dibasic calcium phosphate with sodium carbonate. The product calcium carbonate precipitates leaving dibasic sodium salt in the solution. The solution on cooling yields crystals of hydrated product.
CaHPO4 + Na2CO3 → CaCO3 + Na2HPO4 | | Definition | ChEBI: Disodium hydrogenphosphate is a sodium phosphate. | | General Description | Sodium phosphate dibasic in combination with sodium phosphate monobasic is used in the preparation of oral sodium phosphate solution (OSPS), which is a purgative used for bowel cleansing before colonoscopy. | | Flammability and Explosibility | Non flammable | | Biochem/physiol Actions | Sodium phosphate dibasic is an important component of running buffer of denaturing gel electrophoresis. It induces glomerular calcification as well as other nephrotic syndrome in Sprague-Dawley rats. | | Safety Profile | Poison by intravenous route. Moderately toxic by intraperitoneal, subcutaneous, and intramuscular routes. Mildly toxic by ingestion. A skin and eye irritant. When heated to decomposition it emits toxic fumes of POx and Na2O. See also PHOSPHATES. | | Purification Methods | Crystallise the salt twice from warm water, by cooling. Dry in air, then in an oven overnight at 130o. It should be dried before use as it is slightly hygroscopic. It forms di-, hepta-and deca-hydrates. |
| | Sodium Phosphate, Dibasic Preparation Products And Raw materials |
|