| Company Name: |
Capot Chemical Co.,Ltd. |
| Tel: |
571-85586718 +8613336195806 |
| Email: |
sales@capotchem.com |
| Products Intro: |
Product Name:(3aR,4r,6r,6ar)-4-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-6-(hydroxymethyl)-2,2-dimethyl-6,6a-dihydro-3ah-furo[3,4-d][1,3]dioxole-4-carbonitrile
CAS:1191237-80-5
Purity:98% Min. Package:1G;1KG;100KG
|
|
|
|
|
Remdesivir N-2 manufacturers
- Remdesivir N-2
-

- $80.00 / 1kg
-
2023-09-11
- CAS:1191237-80-5
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 10000tons
- Remdesivir N-2
-

- $180.00 / 1KG
-
2022-09-12
- CAS:1191237-80-5
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 9000kg/per week
|
| | Remdesivir N-2 Basic information |
| Product Name: | Remdesivir N-2 | | Synonyms: | (3aR,4R,6R,6aR)-4-(4-Aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-6-(hydroxymethyl)-2,2-dimethyltetrahydrofuro[3,4-d][1,3]dioxole-4-carbonitrile;Remdesivir Intermediate 4;Remdesivir Related Compound 7;Remdesivir-015;Remdesivir O-desphosphate acetonide impurity;(3αR,4R,6R,6αR)-4-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-6-(hydroxymethyl)-2,2-dimethyltetrahydrofuro[3,4-d][1,3]dioxole-4-carbonitrile;(3Ar,4R,6R,6aR)-4-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-6-(hydroxymethyl)-2,2-dimethyl-6,6a-dihydro-3aH-furo[3,4-d][1,3]dioxole-4-carbonitrile;Remdesivir No. 5 Intermediate | | CAS: | 1191237-80-5 | | MF: | C15H17N5O4 | | MW: | 331.33 | | EINECS: | | | Product Categories: | PEG | | Mol File: | 1191237-80-5.mol | 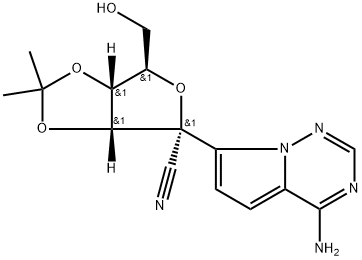 |
| | Remdesivir N-2 Chemical Properties |
| density | 1.66±0.1 g/cm3(Predicted) | | solubility | DMSO:55.5(Max Conc. mg/mL);167.51(Max Conc. mM)
Ethanol:5.0(Max Conc. mg/mL);15.09(Max Conc. mM)
PBS (pH 7.2):0.3(Max Conc. mg/mL);0.91(Max Conc. mM)
DMF:12.0(Max Conc. mg/mL);36.22(Max Conc. mM) | | form | A solid | | pka | 14.14±0.10(Predicted) |
| | Remdesivir N-2 Usage And Synthesis |
| Uses | Remdesivir N-2 can be used as organic synthesis intermediates and pharmaceutical intermediates, mainly used in the research and development and chemical production of Remdesivir. | | Synthesis | 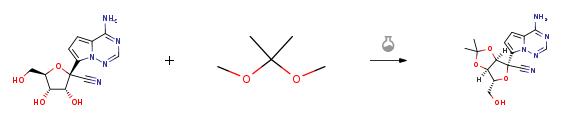
5.62g of compound GS-441524 was dissolved in 30mL of acetone, 11.50mL of 2,2-dimethoxypropane and 1.34mL of sulfuric acid were added, stirred at 45??C for half an hour, cooled to 25??C, and the organic solvent was removed by rotary evaporation. It was extracted with 100 mL of ethyl acetate and 100 mL of saturated sodium bicarbonate aqueous solution, and the extraction was repeated three times. The ethyl acetate layers were combined, dried by adding anhydrous sodium sulfate, and filtered to remove sodium sulfate. The organic solvent was removed by rotary evaporation, and column chromatography was performed (eluent: petroleum ether/ethyl acetate (V/V)=1/2) to obtain 6.20 g of compound 5 (white solid, 97% yield). |
| | Remdesivir N-2 Preparation Products And Raw materials |
| Raw materials | GS-441524-->D-Ribofuranose, 1-C-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-2,3,5-tris-O-(phenylmethyl)--->(3R,4R,5R)-2-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-3,4-bis(benzyloxy)-5-((benzyloxy)methyl)tetrahydrofuran-2-carbonitrile-->Remdesivir impurity2-->2,3,5-TRI-O-BENZYL-L-ARABINOFURANOSE-->7-Iodopyrrolo[2,1-f][1,2,4]triazin-4-amine-->PYRROLO[1,2-F][1,2,4]TRIAZIN-4-AMINE-->2,2-Dimethoxypropane | | Preparation Products | Remdesivir |
|