|
|
| | 4-NITROPYRENE Basic information |
| Product Name: | 4-NITROPYRENE | | Synonyms: | 4-NITROPYRENE;4-nitro-pyren;4-Nitropyrene>;Pyrene, 4-nitro-;Trofinetide Impurity 12 | | CAS: | 57835-92-4 | | MF: | C16H9NO2 | | MW: | 247.25 | | EINECS: | | | Product Categories: | | | Mol File: | 57835-92-4.mol | 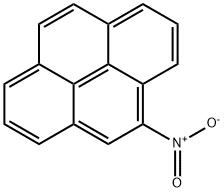 |
| | 4-NITROPYRENE Chemical Properties |
| Melting point | 187.0 to 192.0 °C | | Boiling point | 390.29°C (rough estimate) | | density | 1.1699 (rough estimate) | | refractive index | 1.5300 (estimate) | | storage temp. | -20°C Freezer | | solubility | Acetonitrile (Slightly, Sonicated), Chloroform (Slightly) | | form | Solid | | color | Orange needles from Me2CO/MeOH | | CAS DataBase Reference | 57835-92-4 | | IARC | 2B (Vol. 46, 105) 2014 | | EPA Substance Registry System | 4-Nitropyrene (57835-92-4) |
| | 4-NITROPYRENE Usage And Synthesis |
| Chemical Properties | 4-Nitropyrene is a nitro-substituted polycyclic aromatic hydrocarbonthat exists as orange needles at room temperature (IARC 1989). It is practically insoluble in water but soluble in organic solvents such as acetone, benzene, dimethyl sulfoxide, and methylene chloride (WHO2003). | | Uses | 4-Nitropyrene is used only as a laboratory chemical (IARC 1989); there is no evidence that it has ever been used for commercial purposes. | | Uses | 4-Nitropyrene is a polycyclic aromatic hydrocarbon (PAH) pollutant that was found in ambient air of the Arctic town Longyearbyen, Svalbard. | | Definition | ChEBI: 4-Nitropyrene is a member of pyrenes. | | Safety Profile | Confxmed carcinogen
with experimental carcinogenic data.
Mutation data reported. When heated to
decomposition it emits toxic fumes of NOx. | | Carcinogenicity | 4-Nitropyrene is reasonably anticipated to be a human carcinogenbased on sufficient evidence of carcinogenicity from studies in experimental animals. |
| | 4-NITROPYRENE Preparation Products And Raw materials |
|