| | Antimycin A3 Basic information |
| Product Name: | Antimycin A3 | | Synonyms: | antimycina3;ioxonan-3-yl)-3-formamidosalicylamide,stereoisomer;isovalericacid,n-esterwithn-(7-butyl-8-hydroxy-4,9-dimethyl-2,6-dioxo-1,5-d;[2R-(2R*,3S*,6S*,7R*,8R*)]-8-butyl-3-(3-formamidosalicylamido)-2,6-dimethyl-4,9-dioxo-1,5-dioxonan-7-yl isovalerate;Blastmycin;3-Methyl-butyric acid (2R,3S,6S,7R,8R)-8-butyl-3-{[1-(3-formylamino-2- hydroxy-phenyl)-methanoyl]-amino}-2,6-dimethyl-4,9-dioxo-[1,5]dioxonan -7-yl ester;A 80021F34;Aids012099 | | CAS: | 522-70-3 | | MF: | C26H36N2O9 | | MW: | 520.57 | | EINECS: | 208-335-6 | | Product Categories: | | | Mol File: | 522-70-3.mol | 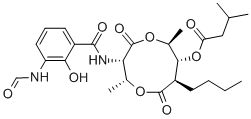 |
| | Antimycin A3 Chemical Properties |
| Melting point | 170.5-171.5° (Liu); 174-174.5° (Kinoshita) | | alpha | D26 +64.3° (chloroform) (Liu); D24 +80° (Kinoshita) | | Boiling point | 603.87°C (rough estimate) | | density | 1.2829 (rough estimate) | | refractive index | 1.5300 (estimate) | | storage temp. | −20°C | | solubility | DMF: Soluble; DMSO: Soluble; Ethanol: Soluble; Methanol: Soluble | | form | A solid | | CAS DataBase Reference | 522-70-3 |
| Hazard Codes | T | | Risk Statements | 23/24/25 | | Safety Statements | 36/37/39-45 | | RIDADR | 3172 | | RTECS | NY1502900 | | HazardClass | 6.1(a) | | PackingGroup | II | | Toxicity | LD50 in mice (mg/kg): 1.8 i.p.; 1.6 s.c. (Watanabe) |
| | Antimycin A3 Usage And Synthesis |
| Chemical Properties | Antimycin (A3C26H36N2O9) and (Antimycin
A1) C28H40N2O9 are crystalline solids. | | Uses | Antimycin A3 is a homologue of the antimycin complex, first isolated in 1958 from Streptomyces blastimyceticus and co-produced with antimycin A4. Antimycin A3 is a more polar analogue of A1 and A2. Like all antimycins, antimycin A3 is a potent inhibitor of respiration by inhibiting the oxidation of ubiquinol to ubiquinone. Antimycin A3 exhibits broad biological activity as an antifungal, anthelmintic, insecticidal, antiviral and antitumor active. Lack of availability of the individual antimycin components has hindered greater understanding of the common and unique molecular targets of the components of the antimycin complex. | | Definition | ChEBI: Purothionin AII is an amidobenzoic acid. | | Potential Exposure | Specific uses for antimycin A were
not found, however, antimycin A1, and antimycin A3 are
reported to be antibiotic substances produced by streptomyces
for use as a fungicide, possible insecticide and miticide.
Registered as a pesticide in the U.S. | | Shipping | UN2811 Toxic solids, organic, n.o.s., Hazard
Class: 6.1; Labels: 6.1-Poisonous materials, Technical
Name Required. |
| | Antimycin A3 Preparation Products And Raw materials |
|