|
|
| | (+)-1,2-BIS[(2S,5S)-2,5-DIMETHYLPHOSPHOLANO]BENZENE Basic information | | Reaction |
| Product Name: | (+)-1,2-BIS[(2S,5S)-2,5-DIMETHYLPHOSPHOLANO]BENZENE | | Synonyms: | (+)-1,2-Bis((2S,5S)-2,5-diMethylphospholano)benzene (S,S)-Me-DUPHOS;(+)-1,2-Bis[(2S,5S)-2,5-diMethylphospholano]benzene kanata purity;(S,S)-METHYL-DUPHOS;(+)-1,2-Bis((2S,5S)-2,5-dimethylphospholano)benzene,98+%(S,S)-Me-DUPHOS;(+)-1,2-BIS[(2S,5S)-2,5-DIMETHYLPHOSPHOLANO]BENZENE;(2S,2'S,5S,5'S)-2,2',5,5'-TETRAMETHYL-1,1'-(1,2-PHENYLENE)DIPHOSPHOLANE;(2S,2'S,5S,5'S)-2,2',5,5'-TETRAMETHYL-1,1'-(O-PHENYLENE)DIPHOSPHOLANE;(+)-1,2-Bis[(2S,5S)-2,5-dimethylphospholano]benzene,(2S,2′S,5S,5′S)-2,2′,5,5′-Tetramethyl-1,1′-(o-phenylene)diphospholane, (S,S)-Me-DUPHOS, (S,S)-Methyl-DUPHOS | | CAS: | 136735-95-0 | | MF: | C18H28P2 | | MW: | 306.36 | | EINECS: | | | Product Categories: | organophosphine ligand | | Mol File: | 136735-95-0.mol | ![(+)-1,2-BIS[(2S,5S)-2,5-DIMETHYLPHOSPHOLANO]BENZENE Structure](CAS/GIF/136735-95-0.gif) |
| | (+)-1,2-BIS[(2S,5S)-2,5-DIMETHYLPHOSPHOLANO]BENZENE Chemical Properties |
| Melting point | 67-76 °C | | alpha | D25 +476± 5° (c = 1 in hexane) | | Boiling point | 415.0±15.0 °C(Predicted) | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | form | crystal | | color | white | | Water Solubility | Insoluble in water. | | Sensitive | Air Sensitive | | BRN | 4810602 |
| | (+)-1,2-BIS[(2S,5S)-2,5-DIMETHYLPHOSPHOLANO]BENZENE Usage And Synthesis |
| Reaction |
- The DUPHOS family of catalysts is highly efficient for the asymmetric hydrogenation of various substituted acetamidoacrylates and enol acetates yielding products of high enantiomeric excesses. Efficient ligand for the asymmetric hydrogenation of imines, enamines, and enamides.
- Asymmetric hydrogenation of vinyl alcohols.
- Catalyst used for the asymmetric hydrogenation of enol phosphonates.
- Asymmetric hydrogenation of allylic alcohols.
- Ligand for the catalytic asymmetric [4+1] cycloaddition of vinylallenes with CO.
- Ligand for the Rh-catalyzed asymmetric enyne cycloisomerization.
- Catalytic enantioselective addition of dialkylzinc to N-Diphenylphosphinoylimines.
- Palladium-catalyzed asymmetric phosphination.
- Palladium-catalyzed asymmetric hydrogenation of carbonyls.
- Palladium-catalyzed 1,4 arylation of α, β-unsaturated ketones.
- Asymmetric, Ir-catalyzed, [2+2+2] cycloaddition.
- Asymmertric palladium-catalyzed synthesis of 2-methyl-indolines via C–H activation.
- Copper-catalyzed monoborylation of 1,3-Dienes.
- Rhodium-catalyzed enantioselective transmetalation.
- CuH-catalyzed hydroamination of styrenes.
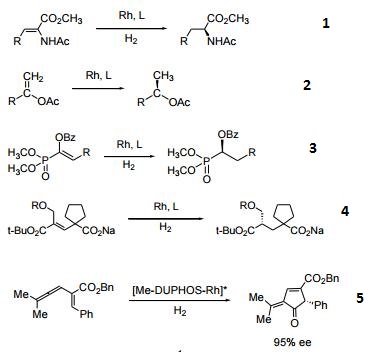
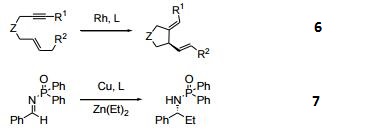
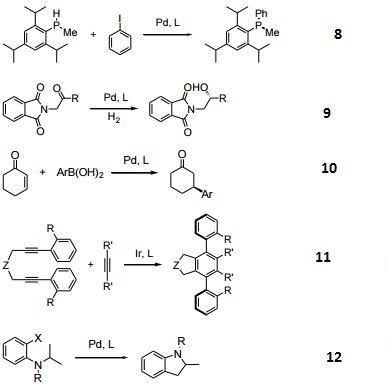
| | Uses | It is a DuPhos and BPE ligands which are highly efficient privileged ligands. |
| | (+)-1,2-BIS[(2S,5S)-2,5-DIMETHYLPHOSPHOLANO]BENZENE Preparation Products And Raw materials |
|