|
|
| | trimercury dioxide sulphate Basic information |
| Product Name: | trimercury dioxide sulphate | | Synonyms: | trimercury dioxide sulphate;Basic mercuric sulfate;Mercuric dioxysulfate;Mercuric subsulfate;Mercury oxide sulfate;Mercury oxonium sulfate;Mercury(II) subsulfate;MERCURIC SULFATE, BASIC | | CAS: | 1312-03-4 | | MF: | Hg3O6S | | MW: | 729.8314 | | EINECS: | 215-191-8 | | Product Categories: | | | Mol File: | 1312-03-4.mol | 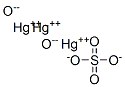 |
| | trimercury dioxide sulphate Chemical Properties |
| | trimercury dioxide sulphate Usage And Synthesis |
| Chemical Properties | yellow powder(s) [CRC10] | | General Description | A lemon-yellow powder or bright yellow scaly material. Very slightly soluble in water and denser than water. Hence sinks in water. Toxic by inhalation and by ingestion. | | Air & Water Reactions | Very slightly soluble in water. | | Reactivity Profile | trimercury dioxide sulphate is soluble in acids. Incompatible with acetylene, ammonia, chlorine dioxide, azides, calcium (amalgam formation), sodium carbide, lithium, rubidium, copper . | | Health Hazard | Highly toxic, may be fatal if inhaled, swallowed or absorbed through skin. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution. | | Fire Hazard | Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Containers may explode when heated. Runoff may pollute waterways. |
| | trimercury dioxide sulphate Preparation Products And Raw materials |
|