|
|
| | NVP-TNKS656 Basic information |
| Product Name: | NVP-TNKS656 | | Synonyms: | NVP-TNKS656;TNKS656;N-(cyclopropylmethyl)-2-(4-(4-methoxybenzoyl)piperidin-1-yl)-N-((4-oxo-3,5,7,8-tetrahydro-4H-pyrano[4,3-d]pyrimidin-2-yl)methyl)acetamide;NVP-TNKS656 - TNKS 656;TNKS656; TNKS-656;TNKS 656;CS-1333;N-(cyclopropylmethyl)-2-(4-(4-methoxybenzoyl)piperidin-1-yl)-N-((4-oxo-4,5,7,8-tetrahydro-3H-pyrano[4,3-d]pyrimidin-2-yl)methyl)acetamide;1-Piperidineacetamide, N-(cyclopropylmethyl)-4-(4-methoxybenzoyl)-N-[(3,5,7,8-tetrahydro-4-oxo-4H-pyrano[4,3-d]pyrimidin-2-yl)methyl]- | | CAS: | 1419949-20-4 | | MF: | C27H34N4O5 | | MW: | 494.58 | | EINECS: | 604-604-1 | | Product Categories: | | | Mol File: | 1419949-20-4.mol | 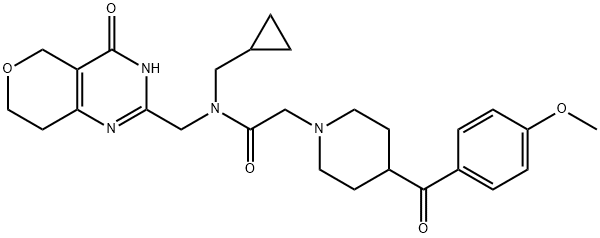 |
| | NVP-TNKS656 Chemical Properties |
| Boiling point | 700.7±70.0 °C(Predicted) | | density | 1.37±0.1 g/cm3(Predicted) | | storage temp. | Sealed in dry,Store in freezer, under -20°C | | solubility | Soluble in DMSO | | form | crystalline solid | | pka | 6.66±0.10(Predicted) |
| | NVP-TNKS656 Usage And Synthesis |
| Description | NVP-TNKS656 is an orally bioavailable inhibitor of tankyrase 2 (TNKS2; IC50 = 6 nM). It is selective for TNKS2 over poly(ADP-ribose) polymerase (PARP) 1 and 2 (IC50s = >19 and 32 nM, respectively). NVP-TNKS656 inhibits Wnt ligand-induced signaling with an IC50 value of 3.5 nM in an HEK293 cell reporter assay. In vivo, NVP-TNKS656 (350 mg/kg) reduces Axin 2 mRNA expression, a Wnt/β-catenin target gene, in MMTV-Wnt1 tumor bearing mice. | | in vitro | the ic50 value of nvp-tnks656 against parp1, tnks2, parp2, and stf was > 19, 0.006, 32, and 0.0035 μm, respectively [1]. | | in vivo | the clearance and volume of distribution of nvp-tnks656 were 10 ml/min/kg and 0.6 l/kg after intravenous administration in mice. the exposure and oral bioavailability was 32% and 53%, respectively. in the mouse mammary tumor virus (mmtv)-wnt1 transgenic model, oral administration of nvp-tnks656 (350 mg/kg) activated the wnt signaling over a 24-h time course. nvp-tnks656 treatment reduced the wnt/beta-catenintarget gene axin2 mrna level by 70-80% [1]. | | references | shultz m d, cheung a k, kirby c a, et al. identification of nvp-tnks656: the use of structure-efficiency relationships to generate a highly potent, selective, and orally active tankyrase inhibitor[j]. journal of medicinal chemistry, 2013, 56(16): 6495-6511. |
| | NVP-TNKS656 Preparation Products And Raw materials |
|