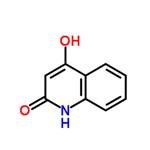
- 2,4-Quinolinediol
-
- $5.00 / 1KG
-
2024-01-14
- CAS:86-95-3
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
- 2,4-Quinolinediol
-

- $30.00/ KG
-
2023-10-08
- CAS:86-95-3
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 20T
- 2,4-Quinolinediol
-

- $100.00 / 1bag
-
2023-09-06
- CAS:86-95-3
- Min. Order: 1bag
- Purity: 99
- Supply Ability: 5000
|
| | 2,4-Quinolinediol Basic information |
| | 2,4-Quinolinediol Chemical Properties |
| Melting point | >300 °C(lit.) | | Boiling point | 287.44°C (rough estimate) | | density | 1.2480 (rough estimate) | | refractive index | 1.5050 (estimate) | | storage temp. | Inert atmosphere,Room Temperature | | solubility | Aqueous Base (Slightly), DMSO (Slightly) | | form | Powder | | pka | 4.50±1.00(Predicted) | | color | Very light brown | | Water Solubility | Insoluble in water | | BRN | 129767 | | InChI | InChI=1S/C9H7NO2/c11-8-5-9(12)10-7-4-2-1-3-6(7)8/h1-5H,(H2,10,11,12) | | InChIKey | HDHQZCHIXUUSMK-UHFFFAOYSA-N | | SMILES | N1C2=C(C=CC=C2)C(O)=CC1=O | | LogP | 1.020 (est) | | CAS DataBase Reference | 86-95-3(CAS DataBase Reference) | | NIST Chemistry Reference | 2,4-Quinolinediol (mainly keto form)(86-95-3) | | EPA Substance Registry System | 2(1H)-Quinolinone, 4-hydroxy- (86-95-3) |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26-37/39 | | WGK Germany | 3 | | RTECS | FG7300000 | | TSCA | Yes | | HazardClass | IRRITANT | | HS Code | 29334900 |
| | 2,4-Quinolinediol Usage And Synthesis |
| Chemical Properties | very light brown powder | | Uses | 2,4-Quinolinediol is a coupling component of yellow azo dyes, also used as pharmaceutical intermediates. | | Application | Researchers have found that the hetarylazoquinoline dyes were prepared by coupling 2,4-quinolinediol with diazotised heterocyclic amines in nitrosylsulphuric acid. In addition, the crude enzyme preparation effectively promotes the reaction between 4-hydroxy-1-methyl-2-quinolinone and 2,4-quinolinediol to afford the corresponding benzofuroquinolinones in satisfactory yields[1-2].
| | Definition | ChEBI: A heteroaryl hydroxy compound that is 2-quinolone substituted at position 4 by a hydroxy group. | | References |
[1] Sener, I. , et al. "Synthesis and investigations of the absorption spectra of hetarylazo disperse dyes derived from 2,4-quinolinediol." DYES AND PIGMENTS (2006).
[2] Angeleska, Simona , P. Kefalas , and A. Detsi . "Crude peroxidase from onion solid waste as a tool for organic synthesis. Part III: synthesis of tetracyclic heterocycles (coumestans and benzofuroquinolinones)." Tetrahedron Letters 54.19(2013):2325-2328.
|
| | 2,4-Quinolinediol Preparation Products And Raw materials |
| Preparation Products | 4-BROMOQUINOLIN-2(1H)-ONE-->sodium [5-chloro-2-hydroxy-3-[(1,2,3,4-tetrahydro-2,4-dioxoquinolin-3-yl)azo]benzene-1-sulphonato(3-)]hydroxychromate(1-)-->DISPERSE YELLOW 56-->Chromium, [3-[[2,3-dihydro-4-hydroxy-2-(oxo-κO)-3-quinolinyl]azo-κN1]-N,N-diethyl-4-(hydroxy-κO)benzenesulfonamidato(3-)]--->Chromate, hydroxy[4-hydroxy-3-nitro-5-[(1,2,3,4-tetrahydro-2,4-dioxo-3-quinolinyl)azo]benzenesulfonato]-, sodium-->Benzenesulfonic acid, 2-[(1,2-dihydro-4-hydroxy-2-oxo-3-quinolinyl)azo]-5-[(4-sulfophenyl)azo]-, disodium salt-->Solvent Yellow 18-->C.I. Acid black 70 |
|