|
|
| | 2,4,6-Triallyloxy-1,3,5-triazi Basic information |
| Product Name: | 2,4,6-Triallyloxy-1,3,5-triazi | | Synonyms: | activatoroc;Cyanuric acid, tri-2-propenyl ester;Perkalink 300;Perkalink 301;perkalink300;perkalink300-50d;rhenofittac;s-Triazine, 2,4,6-tris(allyloxy)- | | CAS: | 101-37-1 | | MF: | C12H15N3O3 | | MW: | 249.27 | | EINECS: | 202-936-7 | | Product Categories: | Chemical | | Mol File: | 101-37-1.mol | 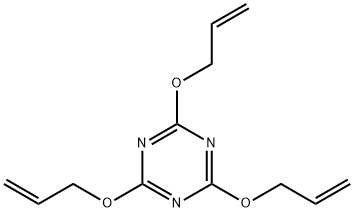 |
| | 2,4,6-Triallyloxy-1,3,5-triazi Chemical Properties |
| Melting point | 26-28 °C | | Boiling point | 156 °C | | density | 1.11 g/mL at 30 °C | | vapor pressure | 1.3 hPa (100 °C) | | refractive index | 1.5290 (estimate) | | Fp | >230 °F | | storage temp. | Store at <= 20°C. | | solubility | 0.3g/l | | pka | 2.04±0.10(Predicted) | | form | Oil | | color | Colourless | | Specific Gravity | 1.11 | | Water Solubility | 6g/L(20 ºC) | | BRN | 235560 | | Stability: | Stable, but moisture-sensitive. Incompatible with acids, peroxides, strong oxidizing agents, copper, iron and its salts. | | LogP | 3.51 at 20℃ | | CAS DataBase Reference | 101-37-1(CAS DataBase Reference) | | NIST Chemistry Reference | 2,4,6-Triallyloxy-1,3,5-triazine(101-37-1) | | EPA Substance Registry System | 1,3,5-Triazine, 2,4,6-tris(2-propenyloxy)- (101-37-1) |
| Hazard Codes | Xn,N | | Risk Statements | 22-51/53 | | Safety Statements | 36-61 | | RIDADR | UN 3077 9/PG 3 | | WGK Germany | 2 | | RTECS | XZ2080000 | | F | 21 | | TSCA | Yes | | HS Code | 2933 69 80 | | HazardClass | 6.1(b) | | PackingGroup | III | | Toxicity | LD50 orally in Rabbit: 753 mg/kg LD50 dermal Rabbit > 2000 mg/kg |
| | 2,4,6-Triallyloxy-1,3,5-triazi Usage And Synthesis |
| Chemical Properties | Colorless liquid or solid. Miscible with acetone,
benzene, chloroform, dioxane, ethyl acetate,
ethanol, and xylene. Combustible. | | Uses | Polymers as monomer and modifier, organic intermediate. Triallyl Cyanurate is used preferentially as a cross-linking agent in copolymers. On heating it may polymerize violently and then isomerize to the more stable triallyl isocyanurate [1025-15-6] with the allyl groups attached to the nitrogen atoms.
At 30 ℃ viscous solutions of prepolymers form slowly. Triallyl cyanurate is used for the production of heat- and solvent-resistant coatings and moldings, reinforced plastics, and adhesives. Addition of 5 – 10% triallyl cyanurate to polyester – styrene or methyl methacrylate yields cast sheets of improved mechanical and thermal stability. Short-term heating of triallyl cyanurate with polymers in the presence of peroxides at 180 ℃ gives cross-linking; valuable copolymers are thus obtained from poly(vinyl chloride) elastomers and fluoroelastomers. Ethylene polymers and copolymers also may be crosslinked under similar reaction conditions. High-impact plastics have been obtained by grafting butyl acrylate-triallyl cyanurate copolymer with a styrene – acrylonitrile mixture. Other examples of peroxide-initiated curing with triallyl cyanurate (2 – 5 %) at 150 – 160 ℃ are polyurethanes, nylons, cellulose, polyoxyethylene, vinyl-substituted polysiloxanes, and acrylate copolymers. Crosslinking of polycarbonate with triallyl cyanurate by UV irradiation in the presence of polythiols gives scratch-resistant coatings. | | Definition | ChEBI: 2,4,6-triallyloxy-1,3,5-triazine is a member of 1,3,5-triazines and an aromatic ether. | | Hazard | Toxic by ingestion and inhalation. | | Flammability and Explosibility | Non flammable | | Safety Profile | Poison by intravenous
route. Flammable when exposed to heat,
flame, or oxidizers. To fight fire, use spray,
foam, dry chemical. When heated to
decomposition or on contact with acid or
acid fumes it emits hghly toxic fumes of
CNand NOx. See also ESTERS and ALLYL,
COMPOUNDS. |
| | 2,4,6-Triallyloxy-1,3,5-triazi Preparation Products And Raw materials |
|