| Company Name: |
J & K SCIENTIFIC LTD.
|
| Tel: |
010-82848833 400-666-7788 |
| Email: |
jkinfo@jkchemical.com |
| Products Intro: |
Product Name:RofluMilast-d4 N-Oxide
CAS:751-97-3
Package:10Mg,1Mg
|
|
| | ROLITETRACYCLINE Basic information |
| Product Name: | ROLITETRACYCLINE | | Synonyms: | 12,12a-pentahydroxy-6-methyl-1,11-dioxo-n-(1-pyrrolidinylmethyl)-1;bristacin;n-(1-pyrrolidinylmethyl)-tetracycline;n-(pyrrolidinomethyl)tetracycline;pirrolidinometil-tetraciclina;prm-tc;pyrrolidinomethyl-tetracyclin;pyrrolidino-methyl-tetracycline | | CAS: | 751-97-3 | | MF: | C27H33N3O8 | | MW: | 527.57 | | EINECS: | 212-031-9 | | Product Categories: | PAVULON | | Mol File: | 751-97-3.mol | 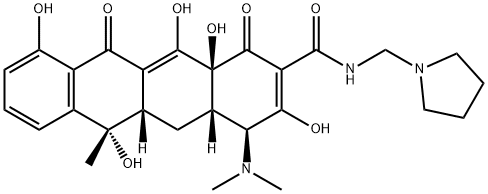 |
| | ROLITETRACYCLINE Chemical Properties |
| Melting point | 163.5°C | | Boiling point | 608.16°C (rough estimate) | | density | 1.2902 (rough estimate) | | refractive index | 1.7120 (estimate) | | storage temp. | -20°C Freezer, Under inert atmosphere | | solubility | DMSO (Slightly), Methanol (Very Slightly) | | form | Solid | | pka | pKa 7.4 (Uncertain) | | color | Very Dark Red to Very Dark Brown | | Water Solubility | >20g/L(21 ºC) |
| Hazard Codes | Xn | | Risk Statements | 22-36/37/38 | | Safety Statements | 26-36 | | WGK Germany | 1 | | RTECS | QI9150000 |
| | ROLITETRACYCLINE Usage And Synthesis |
| Description | Rolitetracycline was synthesized in 1958. Bristol-Myers Laboratories prepared it from tetracycline by introducing a pyrrolidinylmethyl moiety. This antibiotic is very soluble in water and more stable than tetracycline under acidic conditions. Rolitetracycline is used by intravenous injection, and its nitrate is used by both intravenous and intramuscular injections for therapy of the same infections as those treated by tetracycline. | | Uses | Rolitetracycline is a tetracycline antibiotic that may be given intravenously or intramuscularly in serious bacterial infections when oral administration is not practicable. | | Uses | Rolitetracycline, launched in the late 1950s, was the first of the semi-synthetic tetracyclines. Rolitetracycline is formed by a Mannich condensation of formaldehyde and pyrrolidine with tetracycline. Rolitetracycline is a pro-drug of tetracycline, in which the pyrrolidine moiety improves bioavailability compared with tetracycline. Rolitetracycline has broad spectrum Gram positive activity in vivo, but pH instability limits use to parenteral administration. The intrinsic in vitro activity and SARs for this region of the tetracycline molecule have not been extensively investigated. | | Uses | neuromuscular blocker | | Definition | ChEBI: A derivative of tetracycline in which the amide function is substituted with a pyrrolidinomethyl group. | | Brand name | Syntetrin (Bristol-Myers Squibb). | | General Description | Rolitetracycline, N-(pyrrolidinomethyl)tetracycline(Syntetrin), was introduced for use by intramuscular or intravenousinjection. This derivative is made by condensingtetracycline with pyrrolidine and formaldehyde in the presenceof tert-butyl alcohol. It is very soluble in water (1 g dissolvesin about 1 mL) and provides a means of injecting theantibiotic in a small volume of solution. It has been recommendedfor cases when the oral dosage forms are not suitable,but it is no longer widely used. | | Pharmaceutical Applications | 2-N-pyrrolidinomethyl-tetracycline. A semisynthetic derivative
of tetracycline supplied as the nitrate sesquihydrate for
parenteral use.
It is not absorbed from the gastrointestinal tract. It is highly
soluble and therefore can be administered parenterally. Peak
plasma concentrations of 4–6 mg/L occur at 0.5–1 h after
350 mg intravenously. The plasma elimination half-life is 5–8 h.
About 50% of the dose is excreted in the urine, producing
high concentrations.
Intravenous administration is occasionally accompanied by
abnormal taste, shivering and rigors, hot flushes, facial reddening,
dizziness and, rarely, circulatory collapse. Symptoms
of myasthenia gravis have occasionally been exacerbated. |
| | ROLITETRACYCLINE Preparation Products And Raw materials |
|