|
|
| | Etifoxine Basic information |
| Product Name: | Etifoxine | | Synonyms: | etafenoxine;36-801;6-Chloro-2-ethylamino-4-methyl-4-phenyl-4H-3,1-benzoxazine;Etifoxin;HOE-36801;HOE-36-801;2-Ethylamino-6-chloro-4-methyl-4-phenyl-4H-3,1-benzoxazine;Etifoxine | | CAS: | 21715-46-8 | | MF: | C17H17ClN2O | | MW: | 300.78 | | EINECS: | 630-330-6 | | Product Categories: | | | Mol File: | 21715-46-8.mol | 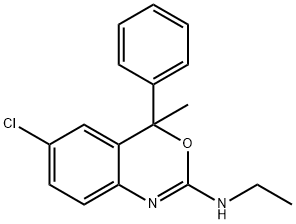 |
| | Etifoxine Chemical Properties |
| Melting point | 90-92℃ | | Boiling point | 421.2±55.0 °C(Predicted) | | density | 1.1716 (rough estimate) | | vapor pressure | 0-1Pa at 20-25℃ | | refractive index | 1.5800 (estimate) | | storage temp. | Store at -20°C | | solubility | DMSO:60.0(Max Conc. mg/mL);199.47(Max Conc. mM)
DMF:30.0(Max Conc. mg/mL);99.74(Max Conc. mM)
DMF:PBS (pH 7.2) (1:8):0.1(Max Conc. mg/mL);0.33(Max Conc. mM)
Ethanol:20.0(Max Conc. mg/mL);66.49(Max Conc. mM) | | form | Solid:particulate/powder | | pka | 4.89±0.40(Predicted) | | LogP | 4.09-4.52 at 21-25℃ and pH7 |
| Toxicity | LD50 orally in mice: 12 g/kg (Hoffmann) |
| | Etifoxine Usage And Synthesis |
| Originator | Circupon,Troponwerke,W. Germany | | Definition | ChEBI: Etifoxine is a benzoxazine. | | Manufacturing Process | 30 parts of 1-(3'-hydroxyphenyl)-2-(N-benzylaminomethyl)-ethan-1-one are
mixed with 100 parts of pyridine and 30 parts of pivalic anhydride and
dissolved while warming. After heating for 1 hour under reflux, the acylation
is complete. After concentrating the reaction solution, the product is
precipitated from acetone/ether. Yield: 96.4% of 1-(3'-pivaloyloxyphenyl)-2-
(N-benzylaminomethyl)-ethan-1-one.
3 parts of palladium/charcoal (10% strength) are prehydrogenated in water,
thereafter 10 parts of 1-(3'-pivaloyloxyphenyl)-2-(N-benzylaminoethyl)-ethan-
1-one, dissolved in a 10-fold amount of water, are added dropwise at room
temperature and hydrogenation is carried out until 1 mol of hydrogen has
been taken up. After filtering off the catalyst, a further 3 parts of
palladium/charcoal are added and hydrogenation is carried out until a further
mol of hydrogen has been taken up. The catalyst is separated off and after
removal of the solvent the hydrogenation product is reprecipitated from
acetone/petroleum ether and from methanol/ether until it is pure according to
thin layer chromatography. Yield: 38.8% of 1-(3'-pivaloyloxyphenyl)-2-
ethylaminoethanol-1 hydroxide, melting point 208°C to 209°C. | | Therapeutic Function | Tranquilizer |
| | Etifoxine Preparation Products And Raw materials |
|