L-Threonolactone manufacturers
- L-Threonolactone
-

- $0.00 / 10G
-
2023-02-16
- CAS:21730-93-8
- Min. Order: 10G
- Purity: 98%min
- Supply Ability: 30kg/month
|
| | L-Threonolactone Basic information |
| Product Name: | L-Threonolactone | | Synonyms: | 3,4-bis(trimethylsilyloxy)oxolan-2-one;Threonolactone;L-Threonic acid-1,4-lactone;(3R,4S)-3,4-Dihydroxydihydrofuran-2(3H)-one;L-Threonic acid γ-lactone;(3S,4S)-3-Amino-4-hydroxydihydrofuran-2(3H)-one;L-Threonic Acid-4-lactone;2(3H)-Furanone, dihydro-3,4-dihydroxy-, (3R,4S)- | | CAS: | 21730-93-8 | | MF: | C4H6O4 | | MW: | 118.09 | | EINECS: | | | Product Categories: | | | Mol File: | 21730-93-8.mol | 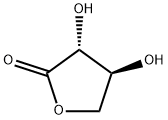 |
| | L-Threonolactone Chemical Properties |
| Melting point | 74-75 °C | | Boiling point | 145-150 °C(Press: 0.25 Torr) | | density | 1.681±0.06 g/cm3(Predicted) | | solubility | Acetonitrile (Sparingly), DMSO (Slightly), Methanol (Sparingly) | | form | Solid | | pka | 12.34±0.40(Predicted) | | color | White to Off-White | | optical activity | [α]/D 29.5±3.5°, c = 1 in H2O | | BRN | 81074 |
| | L-Threonolactone Usage And Synthesis |
| Uses | L-Threonolactone is formed in the autoxidation of L-Ascorbic acid (A786990), a compound that is essential for numerous enzymatic reactions in the body and is also responsible for scavenging free radicals to prevent oxidative damage to cells. | | Definition | ChEBI: A butan-4-olide that is dihydrofuran-2-one substituted at C-3 and C-4 by hydroxy groups (the 3R,4S-diastereomer). |
| | L-Threonolactone Preparation Products And Raw materials |
|