| Company Name: |
J & K SCIENTIFIC LTD.
|
| Tel: |
010-82848833 400-666-7788 |
| Email: |
jkinfo@jkchemical.com |
| Products Intro: |
Product Name:Dimethylammonium dichlorotri(mu-chloro)bis[(S)-(-)-2,2'-bis(diphenylphosphino)-1,1'-binaphthyl]diruthenate(II) [NH2Me2][{RuCl((S)-binap)}2(mu-Cl)3]
CAS:199541-17-8
Package:1g;250mg;5g
|
| Company Name: |
TCI (Shanghai) Development Co., Ltd.
|
| Tel: |
021-67121386 |
| Email: |
Sales-CN@TCIchemicals.com |
| Products Intro: |
Product Name:[NH2Me2][(RuCl((S)-binap))2(mu-Cl)3]
CAS:199541-17-8
Package:1G,200MG
|
|
| | Dimethylammoniumdichlorotri(mu-chloro)bis[(S)-(-)-2,2'-bis(diphenylphosphino)-1,1'-binaphthyl]diruthenate(II) Basic information | | Reaction |
| Product Name: | Dimethylammoniumdichlorotri(mu-chloro)bis[(S)-(-)-2,2'-bis(diphenylphosphino)-1,1'-binaphthyl]diruthenate(II) | | Synonyms: | [NH2Me2][{RuCl((S)-binap)}2(mu-Cl)3];(S)-[(RuCl(BINAP))2(mu-Cl)3][NH2Me2];[NH2ME2][(RUCL((S)-BINAP))2(Μ-CL)3];-binaphthyl]diruthenate(II),[NH2Me2][{RuCl((S);-Cl)3][NH2Me2];DIMETHYLAMMONIUM DICHLOROTRI(Μ-CHLORO)BIS[(S)-(-)-2,2'-BIS(DIPHENYLPHOSPHINO)-1,1'-BINAPHTHYL]DIRUTHENATE(II),[NH2ME2][{RUCL((S);Dimethylammoniumdichlorotri(mu-chloro)bis[(S)-(-)-2,2'-bis(diphenylphosphino)-1,1'-binaphthyl]diruthenate(II);(S)-[(RuCl(BINAP))2(μ-Cl)3][NH2Me2] | | CAS: | 199541-17-8 | | MF: | 2C44H32P2.C2H7N.Cl.Cl3Ru2.ClH | | MW: | 1670.87 | | EINECS: | | | Product Categories: | Ru | | Mol File: | 199541-17-8.mol | ![Dimethylammoniumdichlorotri(mu-chloro)bis[(S)-(-)-2,2'-bis(diphenylphosphino)-1,1'-binaphthyl]diruthenate(II) Structure](CAS/GIF/199541-17-8.gif) |
| | Dimethylammoniumdichlorotri(mu-chloro)bis[(S)-(-)-2,2'-bis(diphenylphosphino)-1,1'-binaphthyl]diruthenate(II) Chemical Properties |
| Melting point | >100°C | | storage temp. | 2-8°C | | Water Solubility | Insoluble in water | | form | Powder | | color | orange | | Sensitive | air sensitive |
| Hazard Codes | F | | Risk Statements | 11 | | RIDADR | UN 1325 4.1/PG 3 | | WGK Germany | 3 | | HS Code | 2843.90.0000 |
| | Dimethylammoniumdichlorotri(mu-chloro)bis[(S)-(-)-2,2'-bis(diphenylphosphino)-1,1'-binaphthyl]diruthenate(II) Usage And Synthesis |
| Reaction |
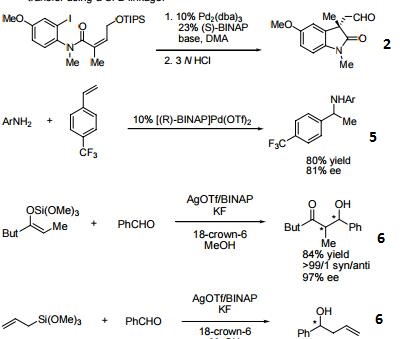
- (R)-BINAP or (R)-Tol-BINAP can be combined with dichloro(1,5-cyclooctadiene)ruthenium to form precursors to NOYORI CATALYST SYSTEMS. These systems exhibit very high catalytic activity and enantioselectivity in the hydrogenation of a wide range of substrates. NOYORI CATALYST SYSTEMS have been shown to effect highly enantioselective hydrogenation of functionalized ketones where the substituents are dialkylamino, hydroxy, siloxy, carbonyl, ester, amide or thioester.
- Useful ligand in asymmetric Heck processes.
- 3. Ligand employed in palladium-catalyzed asymmetric arylation of ketones.
- 4. Ligand employed in rhodium-catalyzed 1,4-additions to enones.
- 5. Ligand employed in palladium-catalyzed hydroamination of styrene derivatives.
- 6. Ligand employed in silver-catalyzed asymmetric Sakuri-Hosomi allylation and Mukaiyama aldol reaction.
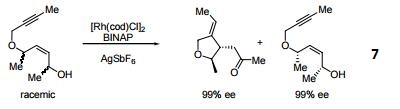
- 7. Ligand employed in rhodium-catalyzed kinetic resolution of enynes.
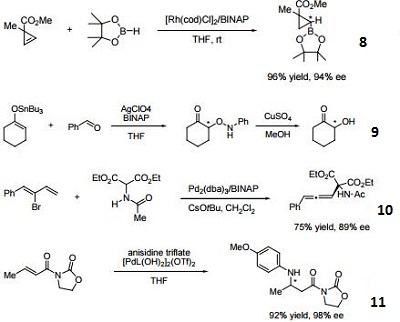
- 8. Ligand employed in asymmetric rhodium-catalyzed hydroboration of cyclopropenes.
- 9. Ligand employed in silver-catalyzed a-hydroxylation of stannyl enol ethers.
- 10. Ligand employed in palladium-catalyzed synthesis of chiral allenes.
- 11. Ligand for palladium-catalyzed enantioselective hetero Michael addition to form b-amino acid derivatives.
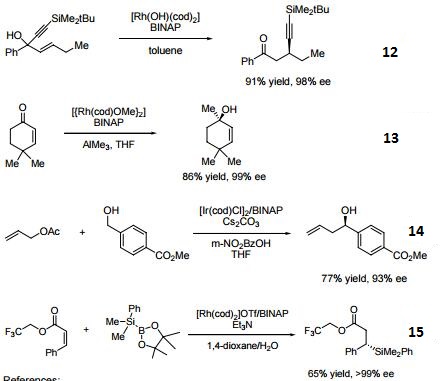
- 12. Ligand employed in rhodium-catalyzed asymmetric rearrangement of alkynyl alkenyl carbinols.
- 13. Ligand employed in rhodium-catalyzed 1,2-addition of aluminium organyl compounds to cyclic enones.
- 14. Ligand employed in iridium-catalyzed transfer hydrogenative allylation of benzylic alcohols.
- 15. Ligand employed in rhodium-catalyzed asymmetric C-Si bond formation by conjugate silyl transfer using a Si-B linkage.
| | Uses | Takasago Ligands and Complexes for Asymmetric Reactions |
| | Dimethylammoniumdichlorotri(mu-chloro)bis[(S)-(-)-2,2'-bis(diphenylphosphino)-1,1'-binaphthyl]diruthenate(II) Preparation Products And Raw materials |
|