| Identification | More | [Name]
Ethynyl estradiol | [CAS]
57-63-6 | [Synonyms]
1,3,5(10)-ESTRATRIEN-17-ALPHA-ETHYNYL-3,17-BETA-DIOL
17a-ethynyl-1,3,5(10)-estratriene-3,17b-diol
17 A-ETHYNYLESTRADIOL
17ALPHA-ETHINYLESTRADIOL
17-ALPHA-ETHYNLESTRADIOL
17ALPHA-ETHYNYL-1,3,5[10]-ESTRATRIENE-3,17BETA-DIOL
17ALPHA-ETHYNYL-DELTA1,3,5(10)-ESTRA-TRIENE-3,17BETA-DIOL
17ALPHA-ETHYNYLESTRADIOL
19-NOR-1,3,5[10],17ALPHA-PREGNATRIEN-20-YNE-3,17-DIOL
19-NORPREGNA-1,3,5(10)-TRIEN-20-YNE-3,17ALPHA-DIOL
ACETENYL ESTRADIOL
ETHINYL ESTRADIOL
ETHYNYLESTRADIOL
LYNORAL
NEO-ESTRONE
NOVESTROL
(17-alpha)-19-norpregna-1,3,5(10)-trien-20-yne-3,17,diol
17-alpha-ethinyl-17-beta-estradiol
17alpha-Ethinyl-17beta-estradiol
17-alpha-ethinyl-3,17-dihydroxy-delta(sup1,3,5)-estratriene | [EINECS(EC#)]
200-342-2 | [Molecular Formula]
C20H24O2 | [MDL Number]
MFCD00003690 | [Molecular Weight]
296.4 | [MOL File]
57-63-6.mol |
| Chemical Properties | Back Directory | [Appearance]
Estradiol, 17-β-is an odorless white to yellow
crystalline substance. | [Melting point ]
182-183 °C(lit.)
| [Boiling point ]
378°C (rough estimate) | [density ]
1.0944 (rough estimate) | [refractive index ]
-30 ° (C=0.4, Pyridine) | [Fp ]
9℃ | [storage temp. ]
room temp | [solubility ]
ethanol: 50 mg/mL, clear, slightly yellow
| [form ]
neat | [pka]
pKa 10.32 (Uncertain) | [color ]
White to Light yellow to Light orange | [Usage]
A synthetic steroid with high oral estrogenic potency | [Merck ]
3734 | [BRN ]
2419975 | [BCS Class]
3/1 | [InChIKey]
BFPYWIDHMRZLRN-SLHNCBLASA-N | [CAS DataBase Reference]
57-63-6(CAS DataBase Reference) | [NIST Chemistry Reference]
Ethinyl estradiol(57-63-6) | [EPA Substance Registry System]
57-63-6(EPA Substance) |
| Safety Data | Back Directory | [Hazard Codes ]
T | [Risk Statements ]
R45:May cause cancer.
R22:Harmful if swallowed. | [Safety Statements ]
S53:Avoid exposure-obtain special instruction before use .
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection .
S45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible) . | [RIDADR ]
UN1230 - class 3 - PG 2 - Methanol, solution | [WGK Germany ]
3
| [RTECS ]
RC8925000
| [F ]
8 | [HS Code ]
29372300 | [Safety Profile]
Confirmed carcinogen
with experimental carcinogenic,
tumorigenic, and neoplastigenic data. Poison
by intraperitoneal route. Moderately toxic by
ingestion. Human systemic effects by
ingestion: glandular effects. An experimental
teratogen. Experimental reproductive
effects. Human mutation data reported.
When heated to decomposition it emits
acrid smoke and irritating fumes. See also
ESTRADIOL | [Hazardous Substances Data]
57-63-6(Hazardous Substances Data) | [Toxicity]
LD50 oral in rat: 960mg/kg |
| Hazard Information | Back Directory | [General Description]
Fine white to creamy white powder. A synthetic steroid. Used in combination with progestogen as an oral contraceptive. | [Reactivity Profile]
ETHINYLESTRADIOL(57-63-6) may react vigorously with strong oxidizing agents. May react exothermically with reducing agents to generate gaseous hydrogen. | [Air & Water Reactions]
Air and light sensitive . Insoluble in water. | [Health Hazard]
ACUTE/CHRONIC HAZARDS: When heated to decomposition this compound emits acrid smoke and fumes. | [Potential Exposure]
The working environment may be
contaminated during sex hormone manufacture, especially
during the extraction and purification of natural steroid hormones; grinding of raw materials; handling of powdered
products and recrystallization. Airborne particles of sex
hormones may be absorbed through the skin, ingested or
inhaled. Enteric absorption results in quick inactivation of
sex hormones in the liver. The rate of inactivation is
decreased for the oral, alkylated steroid hormones (methyl
testosterone, anabolic steroids, etc.). Sex hormones may
accumulate and reach relatively high levels even if their
absorption is intermittent. Consequently, repeated absorption of small amounts may be detrimental to health.
Intoxication by sex hormones may occur in almost all the
exposed workers if preventive measures are not taken. The
effect in the industrial sector is more successful than
the agricultural one (chemical caponizing of cockerels by
stilbestrol implants and incorporation of estrogens in feed
for body weight gain promotion in beef cattle), where measures taken are summary and the number of cases of intoxication is consequently bigger | [Fire Hazard]
The flash point data for this compound are not available. ETHINYLESTRADIOL is probably combustible. | [First aid]
Skin Contact: Flood all areas of body that
have contacted the substance with water. Don’t wait to
remove contaminated clothing; do it under the water stream.
Use soap to help assure removal. Isolate contaminated
clothing when removed to prevent contact by others. Eye
Contact: Remove any contact lenses at once. Immediately
flush eyes well with copious quantities of water or normal
saline for at least 20�30 minutes. Seek medical attention.
Inhalation: Leave contaminated area immediately; breathe
fresh air. Proper respiratory protection must be supplied to
any rescuers. If coughing, difficult breathing or any other
symptoms develop, seek medical attention at once, even if
symptoms develop many hours after exposure. Ingestion:
Contact a physician, hospital or poison center at once. If the
victim is unconscious or convulsing, do not induce vomiting
or give anything by mouth. Assure that the patient’s airway
is open and lay him on his side with his head lower than his
body and transport immediately to a medical facility. If conscious and not convulsion, give a glass of water to dilute
the substance. Vomiting should not be induced without a
physician’s advice | [Shipping]
UN3249 Medicine, solid, toxic, n.o.s., Hazard
Class: 6.1; Labels: 6.1-Poisonous materials | [Incompatibilities]
May react exothermically with reducing
agents to generate flammable gaseous hydrogen.
Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine,
fluorine, etc.); contact may cause fires or explosions.
Keep away from alkaline materials, strong bases, strong
acids, oxoacids, and epoxides. | [Chemical Properties]
Estradiol, 17-β-is an odorless white to yellow
crystalline substance. | [Chemical Properties]
Ethinylestradiol is a white to creamy-white
powder. Odorless. | [Chemical Properties]
Off-White to Light-Yellow Crystalline Powder | [Waste Disposal]
It is inappropriate and possibly dangerous to the environment to dispose of expired or
waste drugs and pharmaceuticals by flushing them down
the toilet or discarding them to the trash. Household quantities of expired or waste pharmaceuticals may be mixed
with wet cat litter or coffee grounds, double-bagged in
plastic, discard in trash. Larger quantities shall carefully
take into consideration applicable DEA, EPA, and FDA
regulations. If possible return the pharmaceutical to the
manufacturer for proper disposal being careful to properly
label and securely package the material. Alternatively, the
waste pharmaceutical shall be labeled, securely packaged
and transported by a state licensed medical waste contractor
to dispose by burial in a licensed hazardous or toxic waste
landfill or incinerator | [Originator]
Estinyl,Schering,US,1944 | [Uses]
A metabolite of 17a-Ethynylestradiol | [Uses]
A synthetic estradiol analog. | [Uses]
A synthetic steroid with high oral estrogenic potency | [Uses]
estrogen, plus progestogen as oral contraceptive | [Definition]
ChEBI: A 3-hydroxy steroid that is estradiol substituted by a ethynyl group at position 17. It is a xenoestrogen synthesized from estradiol and has been shown to exhibit high estrogenic potency on oral administration. | [Manufacturing Process]
In about 250 cc of liquid ammonia (cooled with dry ice and acetone) are
dissolved about 7.5 g of potassium and into the solution acetylene is passed
until the blue color has disappeared (about 3 hours). Then slowly a solution or
suspension of 3 g of estrone in 150 cc of benzene and 50 cc of ether is added.
The freezing mixture is removed, the whole allowed to stand for about 2
hours and the solution further stirred overnight. Thereupon the reaction
solution is treated with ice and water, acidified with sulfuric acid to an acid
reaction to Congo red and the solution extracted five times with ether. The
combined ether extracts are washed twice with water, once with 5% sodium
carbonate solution and again with water until the washing water is neutral.
Then the ether is evaporated, the residue dissolved in a little methanol and
diluted with water. The separated product is recrystallized from aqueous
methanol. The yield amounts to 2.77 g. The 17-ethinyl-estradiol-3,17 thus obtained melts at 142°C to 144°C. | [Brand name]
Estinyl (Schering); Feminone
(Pharmacia & Upjohn); Lynoral (Organon). | [Therapeutic Function]
Estrogen | [Biochem/physiol Actions]
17α-Ethynylestradiol is an orally bio-active synthetic estrogen used as an oral contraceptive. | [Synthesis]
Ethinyl estradiol, 17|á-ethinyl-1,3,5(10)-estratrien-3-17|?-diol (28.1.26),
is made either by condensing estrone with acetylene in the presence of potassium hydroxide
(Favorskii reaction), or by reacting sodium acetylenide in liquid ammonia with estrone.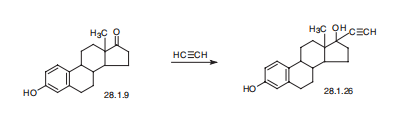
| [storage]
Store at-20°C | [Purification Methods]
17--Ethynylestradiol forms a hemihydrate on recrystallising from MeOH/H2O. It dehydrates on melting and remelts on further heating at m 182-184o. The UV has max at 281nm ( 2040) in EtOH. Its solubility is 17% in EtOH, 25% in Et2O, 20% in Me2CO, 25% in dioxane and 5% in CHCl3. [Petit & Muller Bull Soc Chim Fr 121 1951.] The diacetyl derivative has m 143-144o (from MeOH) and [] D 20 +1o (c 1, CHCl3) [Mills et al. J Am Chem Soc 80 6118 1958]. [Beilstein 6 IV 6877.] |
| Questions And Answer | Back Directory | [Description]
Ethynyl estradiol (EE) is a synthetic form of estrogen that is majorly employed in numerous hormonal contraceptives in combination with progestins. Occasionally, it is also used as a constituent of menopausal hormone therapy with combination with progestins for treating menopausal symptoms. Previously, the drug was used solely for numerous indications such as treatment of prostate cancer and gynecological disorders.
Ethynyl estradiol is a semisynthetic alkylated estradiol with a 17-alpha-ethinyl substitution. It is normally administred orally and is marketed mostly as a combination oral contraceptive under various brand names such as Alesse, Tri-cyclen, Yasmin, and Triphasil. A black box warning is normally labelled on the packaging that states that the drug should not be used in smoking women who are over 35 years old as it can lead to increased risk of serious cardiovascular side effects.
| [History]
Ethynyl estradiol was first developed in 1930 and officially introduced for medical used in 1943. In the 1960s, the drug was widely used in birth control pills. EE is currently found in most combined forms of birth control pills, which makes it one of the most used estrogens.
| [Indications]
Ethynyl estradiol is used for treatment of moderate to severe vasomotor symptoms such as night sweats, hot flashes, and flushing that are associated with the menopause, prostatic carcinoma-palliative therapy of advanced disease, female hypogonadism, as emergency contraceptive, breast cancer, and as an oral contraceptive.
| [Pharmacodynamics]
Ethynyl estradiol is a synthetic derivative of the natural estrogen estradiol. The drug is one of two widely used in oral contraceptive pills. Mestranol is the other drug that is normally converted to ethinyl estradiol before it becomes biologically active. EE is used together with norethindrone as an oral contraceptive agent.
| [Mechanism of Action]
The estrogen in the drug diffuse into their target cells before interacting with a protein receptor. Notably, target cells include the female reproductive tract, the hypothalamus, the mammary gland, and the pituitary. Estrogens act by increasing the hepatic synthesis of thyroid binding globulin (TBG), sex hormone binding globulin (SHBG) as well as other serum proteins. It also acts by suppressing follicle-stimulating hormone (FSH) from anterior pituitary. All these activities are initiated by fist binding to the estrogen receptors. The hypothalamic-pituitary system is suppressed by the combination of an estrogen with a progestin, which decreases the secretion of gonadorotropin-releasing hormone (GnRH).
| [Medical Uses]
Ethinyl Estradiol is majorly used to control pregnancy after sex as a contraception in combined oral contraceptives (COC), which are also referred to as birth control. EE is not only used in preventing pregnancy but is also employed in the treatment of absence of menstruation, acne, as well as symptoms during menstruation. It majorly works by preventing ovulation (the release of an egg) during the menstrual cycle. Further, it acts by making the vaginal fluid thicker to help in preventing sperm from fertilizing the released egg. It also changes the lining of the uterus to prevent attachment of a fertilized egg. EE also works by making the menstruation cycle more regular, decreasing blood loss and painful periods, and decreasing the risk of ovarian cysts.EE is also used as menopausal hormone therapy (HRT) which has numerous benefits such as vaginal itching, vaginal dryness (which normally causes pain during sexual intercourse), and depressed mood. Previously, it was used as a constituent of feminizing hormone therapy for transgender women; however, the use of estradiol has largely superseded it in this therapy. The drug can also be used in preventing osteoporosis and in treating hypogonadism in women and has been used as palliative care for breast cancer in women and prostate cancer in men.
| [Contraindications]
Ethnynyl estradiol should not be prescribed to individuals with history of susceptibility to venous or arterial thrombosis (blood clots) as it can lead to increased cardiovascular problems such as myocardial infarction, venous thromboembolism, and ischemic stroke. As such, it women with acute deep vein thrombosis or pulmonary embolism, any vascular disease history of DVT/PE, and complicated valvular heart disease are not advised to take the drug.
| [Side Effects]
The side effects of EE include vomiting, nausea, headache, tenderness of the breast, abdominal cramps/bloating, swelling of the feet/ankles, and change in weight. In case of severe effects, one should immediately contact the doctor. EE can also cause irregular periods, which is considered normal.
| [Precautions]
Before taking EE, it is important to inform the doctor or pharmacist if you allergic to the drug or to any other estrogens such as mestranol. It is also important to inform the doctor if one has a medical history of blood clots, for instance in the eyes, legs, and lungs. The medication can also affect blood sugar levels if one is diabetic. However, it is important to monitor the blood sugar levels and share it with the doctor in case one is using the drug.
| [Interactions]
Drug interactions may change how EE works or might increase its side effects. Products that can interact with the drug include aromatase inhibitors (such as exemestane and anastrozole), tizanidine, tamoxifen, and tranexamic. It can also interact with certain combination products that are normally used to treat chronic hepatitis C. some drugs interact with EE by decreasing the amount of the drug in the body.
|
|
|