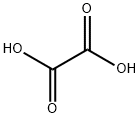
옥살산
|
|
옥살산 속성
- 녹는점
- 189.5 °C (dec.)(lit.)
- 끓는 점
- 365.1°C (estimate)
- 밀도
- 0.99 g/mL at 25 °C
- 증기 밀도
- 4.4 (vs air)
- 증기압
- <0.01 mm Hg ( 20 °C)
- 굴절률
- 1.4261 (estimate)
- 인화점
- 101-157°C
- 저장 조건
- Store below +30°C.
- 용해도
- 물: 25°C에서 용해성108g/L
- 물리적 상태
- 액체
- 산도 계수 (pKa)
- 1.23(at 25℃)
- 색상
- 하얀색
- pH 범위
- 6 - 8 at 25 °C
- 수소이온지수(pH)
- 3(1 mM solution);2.09(10 mM solution);1.31(100 mM solution);
- 냄새
- 냄새 없는
- 수용성
- 90g/L(20℃)
- 승화점
- 101-157 ºC
- Merck
- 14,6911
- BRN
- 385686
- Henry's Law Constant
- 1.43 at pH 4 (quoted, Gaffney et al., 1987)
- 노출 한도
- NIOSH REL: TWA 1, STEL 2, IDLH 500; OSHA PEL: TWA 1; ACGIH TLV: TWA 1, STEL 2 (adopted).
- 안정성
- 안정적이지만 습기에 민감합니다. 금속과 호환되지 않습니다.
- InChIKey
- MUBZPKHOEPUJKR-UHFFFAOYSA-N
- LogP
- -1.7 at 23℃
- CAS 데이터베이스
- 144-62-7(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xn | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 21/22-63-34-41 | ||
| 안전지침서 | 24/25-23-36/37/39-27-26-39-37-36-36/37 | ||
| 유엔번호(UN No.) | UN 3261 8/PG 3 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | RO2450000 | ||
| TSCA | Yes | ||
| 위험 등급 | 8 | ||
| 포장분류 | III | ||
| HS 번호 | 29171110 | ||
| 유해 물질 데이터 | 144-62-7(Hazardous Substances Data) | ||
| 독성 | LD50 orally in Rabbit: 375 mg/kg | ||
| IDLA | 500 mg/m3 | ||
| 기존화학 물질 | KE-13152 |
옥살산 C화학적 특성, 용도, 생산
개요
상온에서는 백색의 고체 상태로 존재한다. 189.5°C에서 분해되며 녹는다. 옥살산 이수화물(C2H2O4·2 H2O)은 101.5°C에 녹는다. 산화되기 쉬워서 정량할 때는 묽은 황산 용액에서 과망가니즈산 칼륨으로 산화시키는 방법을 사용한다. 진한 황산과 함께 가열되면 같은 부피의 일산화 탄소와 이산화 탄소를 생성한다.용도
표백제의 주 성분이며, 포도당과 물엿을 만들 때 쓰이기도 한다. 또한 강한 환원제라 녹 제거나 희토류 추출용으로도 쓴다.안전성
옥살산은 독성이 있기 때문에 취급에 유의해야 한다. 삼킬 경우 치명적일 수 있으며, 부식성이다. 피부, 눈, 기도 등에 심각한 염증이나 화상을 일으킬 수 있고, 피부를 통해 흡수될 수도 있다. 옥살산이 체내에 유입될 경우 신장에 손상을 줄 수 있다.화학적 성질
Oxalic acid is a colorless, odorless powder, or granular solid. The anhydrous form (COOH)2 is an odorless, white solid; the solution is a colorless liquid.물리적 성질
Colorless and odorless rhombic crystals. Hygroscopic. soluble in ethanol, soluble in water, slightly soluble in ether, insoluble in benzene and chloroform.용도
Oxalic acid was used: · in the synthesis of hemicellulose hydrolysates of yellow poplars; · in the synthesis of three-dimensionally ordered macroporous metal oxides or carbonates via templating with polystyrene spheres; · as supporting electrolyte in the electrochemical synthesis of polyaniline-polypyrrole composite coatings.정의
ChEBI: Oxalic acid is an alpha,omega-dicarboxylic acid that is ethane substituted by carboxyl groups at positions 1 and 2. It has a role as a human metabolite, a plant metabolite and an algal metabolite. It is a conjugate acid of an oxalate(1-) and an oxalate.생산 방법
Many industrial processes have been employed for the manufacture of oxalic acid since it was first synthesized. The following processes are in use worldwide: oxidation of carbohydrates, the ethylene glycol process, the propylene process, the dialkyl oxalate process, and the sodium formate process. Sodium formate process is no longer economical in the leading industrial countries, except for China.Nitric acid oxidation is used where carbohydrates, ethylene glycol, and propylene are the starting materials. The dialkyl oxalate process is the newest, where dialkyl oxalate is synthesized from carbon monoxide and alcohol, then hydrolyzed to oxalic acid. This process has been developed by UBE Industries in Japan.Many attempts have been made to synthesize oxalic acid by electrochemical reduction of carbon dioxide in either aqueous or nonaqueous electrolytes.
화학 반응
The reactions of oxalic acid, including the formation of normal and acid salts and esters, are typical of the dicarboxylic acids class. Oxalic acid, however, does not form an anhydride.On rapid heating, oxalic acid decomposes to formic acid, carbon monoxide, carbon dioxide, and water. In aqueous solution, it is decomposed by uv, x-ray, or γ -radiation with the liberation of carbon dioxide. Photodecomposition also occurs in the presence of uranyl salts.
Oxalic acid is a mild reducing agent, and is oxidized by potassium permanganate in acid solution to give carbon dioxide and water. Oxalic acid is catalytically reduced by hydrogen in the presence of ruthenium catalyst to ethylene glycol, and electronically reduced to glyoxylic acid.
Oxalic acid reacts with various metals to form metal salts, which are quite important as the derivatives of oxalic acid. It also reacts easily with alcohols to give esters.
일반 설명
Odorless white solid. Sinks and mixes with water.공기와 물의 반응
Water soluble. Hygroscopic반응 프로필
Oxalic acid is hygroscopic and sensitive to heat. Oxalic acid may react violently with furfuryl alcohol, silver, sodium, perchlorate, sodium hypochlorite, strong oxidizers, sodium chlorite, acid chlorides, metals and alkali metals. . The heating of mixtures of Oxalic acid and urea has lead to explosions. This is due to the rapid generation of the gases, CO2, CO, and NH3, [Praxis Naturwiss. Chem., 1987, 36(8), 41-42]. Oxalic acid and urea react at high temperatures to form toxic and flammable ammonia and carbon monoxide gasses, and inert CO2 gas [Von Bentzinger, R. et al., Praxis Naturwiss. Chem., 1987, 36(8), 41-42].건강위험
As dust or as a solution, can cause severe burns of eyes, skin, or mucous membranes. Ingestion of 5 grams has caused death with symptoms of nausea, shock, collapse, and convulsions coming on rapidly. Repeated or prolonged skin exposure can cause dermatitis and slow-healing ulcers.화재위험
Special Hazards of Combustion Products: Generates poisonous gases농업용
Oxalic acid, (COOH)2, also called ethanedioic acid, is a white, crystalline solid, slightly soluble in water. It is a naturally occurring highly oxidized organic compound with significant chelating activity. It is strongly acidic and poisonous, produced by many plants like sorrel (sourwood), the leaf blades of rhubarb, bark of eucalyptus and many plant roots. In plant cells and tissues, oxalic acid gets accumulated as either sodium, potassium or calcium oxalate, of which the latter occurs as crystals. In turn, salts of oxalic acids enter the bodies of animals and human beings, causing pathological disorders, depending upon the amount consumed. Many species of fungi like Aspergillus, Penicillium, Mucor, as well as some lichens and slime moulds produce calcium oxalate crystals. Upon the death of these microorganisms, plants and animals, the salts get released into the soil, causing some amount of toxicity. However, oxalate-degrading microbes, called Oxalobacter formigenes, decrease oxalate absorption in animals and humans.Oxalic acid is the first of a series of dicarboxylic acids. It is used (a) as a bleaching agent for stains like rust or ink, (b) in textile and leather production, and (c) as monoglyceryl oxalate in the production of ally1 alcohol and formic acid.
잠재적 노출
Oxalic acid is used in textile finishing, paint stripping; metal and equipment cleaning; as an intermediate; as an analytic reagent and in the manufacture of dyes, inks, bleaches, and paint removers; varnishes, wood, and metal cleansers; dextrin, cream of tartar, celluloid, oxalates, tartaric acid, purified methyl alcohol, glycerol, and stable hydrogen cyanide. It is also used in the photographic, ceramic, metallurgic, rubber, leather, engraving, pharmaceutical, paper, and lithographic industries.환경귀착
Biological. Heukelekian and Rand (1955) reported a 5-d BOD value of 0.12 g/g which is 66.7% of the ThOD value of 0.18 g/g.Chemical/Physical. At temperatures greater than 189.5 °C, decomposes to carbon dioxide, carbon monoxide, formic acid, and water (Windholz et al., 1983). Ozonolysis of oxalic acid in distilled water at 25 °C under acidic conditions (pH 6.3) yielded carbon dioxide (Kuo et al., 1977). Absorbs moisture in air forming the dihydrate (Huntress and Mulliken, 1941).
Reacts with bases forming water soluble salts.
운송 방법
UN3261 Corrosive solid, acidic, organic, n.o.s., Hazard class: 8; Labels: 8-Corrosive material, Technical Name Required.비 호환성
The aqueous solution is a medium-strong acid. Compounds of the carboxyl group react with all bases, both inorganic and organic (i.e., amines) releasing substantial heat, water and a salt that may be harmful. Incompatible with arsenic compounds (releases hydrogen cyanide gas), diazo compounds, dithiocarbamates, isocyanates, mercaptans, nitrides, and sulfides (releasing heat, toxic, and possibly flammable gases), thiosulfates and dithionites (releasing hydrogen sulfate and oxides of sulfur). Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from silver compounds; strong alkalis; chlorites. Contact with some silver compounds forms explosive materials.폐기물 처리
Pretreatment involves chemical reaction with limestone or calcium oxide forming calcium oxalate. This may then be incinerated utilizing particulate collection equipment to collect calcium oxide for recycling.옥살산 준비 용품 및 원자재
원자재
일산화탄소(가스)
옥살산
옥살산나트륨
산화 질소
PALLADIUM-CATALYSTS
METALLURGICAL COKE
탄산 납
칼슘 옥살레이트
포름산나트륨
질산
모노에틸렌글리콜
수산화나트륨
감자전분
옥산살디노르말부틸에스테러
황산
준비 용품
7-(트리플루오로메틸)퀴놀린
옥살산,칼륨염
로진
칼륨 비녹살산
암모늄옥살산 제2철
알파Z우린 A
다이메틸옥살레이트
글리세오풀빈
옥살산디에틸
바륨 옥사레이트
니켈(II)옥살산염
Lincomycin hydrochloride monohydrate
옥살산나트륨
옥살산 디암모늄
alpha-(1-Naphthylmethyl)-2-tetrahydrofuranpropionic acid diethylaminoethyl ester oxalate
Filth remover
산화 스칸듐
옥시테트라시클린
3-METHYL-1H-PYRAZOLO[3,4-C]PYRIDAZINE-4,5-DIOL
하이드로퀴논
염화옥살릴
Lubricant
포타슘 타이타늄 옥살레이트
옥산살디노르말부틸에스테러
암모늄 바나딘산염
산화코발트
글리옥실산
옥살산 공급 업체
글로벌( 762)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 |
info@fdachem.com | China | 19368 | 58 |
| Aladdin Scientific | +1-+1(833)-552-7181 |
sales@aladdinsci.com | United States | 57511 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12470 | 58 |
| Hebei Yanxi Chemical Co., Ltd. | +8617531190177 |
peter@yan-xi.com | China | 6011 | 58 |
| Hebei Duling International Trade Co. LTD | +8617333973358 |
sales01@hbduling.cn | China | 15620 | 58 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 |
deasea125996@gmail.com | China | 2503 | 58 |
| Hebei Kingfiner Technology Development Co.Ltd | +86-15532196582 +86-15373005021 |
lisa@kingfinertech.com | China | 3003 | 58 |
| Anhui Zhongda Biotechnology Co., Ltd | +8615689548120 |
linda@zhongda-biotech.com | China | 204 | 58 |
| Ouhuang Engineering Materials (Hubei) Co., Ltd | +8617702722807 |
admin@hbouhuang.com | China | 3001 | 58 |
| Hebei Zhuanglai Chemical Trading Co.,Ltd | +8613343047651 |
admin@zlchemi.com | China | 2999 | 58 |