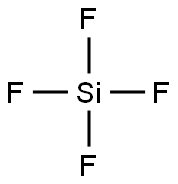실리콘 테트라플루오리드
|
|
실리콘 테트라플루오리드 속성
- 녹는점
- -90°C
- 끓는 점
- -86 °C
- 밀도
- 1.66
- 증기압
- 3517.191kPa at 25℃
- 용해도
- reacts with H2O
- 물리적 상태
- 무색 가스
- 색상
- 무색의
- Specific Gravity
- 1.66
- 수용성
- H2O에 의해 가수분해됨 [AIR87]
- Hydrolytic Sensitivity
- 8: reacts rapidly with moisture, water, protic solvents
- Merck
- 13,8575
- CAS 데이터베이스
- 7783-61-1(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | C,T,T+ | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 26-35-34-31-26/27/28-14 | ||
| 안전지침서 | 7/9-24/25-45-36/37/39-26-23 | ||
| 유엔번호(UN No.) | 1859 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | VW2327000 | ||
| 위험 참고 사항 | Toxic/Corrosive | ||
| TSCA | Yes | ||
| 위험 등급 | 2.3 | ||
| 유해 물질 데이터 | 7783-61-1(Hazardous Substances Data) | ||
| 기존화학 물질 | KE-31045 | ||
| 사고대비 물질 필터링 | 97 |
실리콘 테트라플루오리드 C화학적 특성, 용도, 생산
화학적 성질
Silicon tetrafluoride exhibits a very pungent odor. Silicon tetrachloride has a suffocating odor and is moisture sensitive.물리적 성질
Colorless gas; very pungent odor; fumes heavily in moist air; density of the gas 4.69 g/L; heavier than air, density in air 3.5 (air = 1); sublimes at -95.7°C; solidifies at -90.2°C (under pressure); critical pressure 50atm; decomposes in water forming silicic acid and hydrofluoric acid.용도
Manufacture of fluosilicic acid, intermediate in manufacture of pure silicon, to seal water out of oil wells during drilling.제조 방법
Silicon tetrafluoride is prepared by heating silica with dilute hydrofluoric acid at high temperatures:SiO2 + 4HF → SiF4 + 2H2O
Also, the tetrafluoride may be obtained by heating the elements:
Si + 2F2 → SiF4.
일반 설명
A colorless, nonflammable, corrosive and toxic gas with a pungent odor similar to that of hydrochloric acid. Very toxic by inhalation. Vapor is heavier than air. Under prolonged exposure to heat the containers may rupture violently and rocket.공기와 물의 반응
Fumes in air. Decomposed exothermically by water or moisture in the air to hydrofluoric acid and silicic acid [Merck 11th ed. 1989; Handling Chemicals Safely 1980 p. 821].반응 프로필
When heated to decomposition SILICON TETRAFLUORIDE emits toxic fluoride fumes [Lewis, 3rd ed., 1993, p. 1138]. Reacts violently with alcohols to form HF. Attacks many metals in the presence of moisture. Reacts violently to explosively with lithium nitride [Porritt, L. J., Chem. Brit., 1979, 15, p. 282]. Mixtures with sodium are shock-sensitive explosives [Leleu, Cahiers, 1975, (79), p. 270].위험도
Toxic by inhalation, strong irritant to mucous membranes.건강위험
TOXIC; may be fatal if inhaled, ingested or absorbed through skin. Vapors are extremely irritating and corrosive. Contact with gas or liquefied gas may cause burns, severe injury and/or frostbite. Fire will produce irritating, corrosive and/or toxic gases. Runoff from fire control may cause pollution.화재위험
Some may burn but none ignite readily. Vapors from liquefied gas are initially heavier than air and spread along ground. Some of these materials may react violently with water. Cylinders exposed to fire may vent and release toxic and/or corrosive gas through pressure relief devices. Containers may explode when heated. Ruptured cylinders may rocket.Safety Profile
A poison. Moderately toxic by inhalation. A corrosive irritant to skin, eyes, and mucous membranes. When heated to decomposition it emits toxic fumes of F-. See also FLUORIDES and HYDROFLUORIC ACID잠재적 노출
Silicon tetrafluoride is not used in industry but may be a discharge byproduct of certain processes, including ore refining and smelting operations. It has been used in the manufacture of silane and fluosilicic acid, and pure electronic silicon.운송 방법
UN1859 Silicon tetrafluoride, Hazard Class: 2.3; Labels 2.3-Poisonous gas; 8-Corrosive material; Inhalation Hazard Zone B. Cylinders must be transported in a secure upright position, in a well-ventilated truck. Protect cylinder and labels from physical damage. The owner of the compressed gas cylinder is the only entity allowed by federal law (49CFR) to transport and refill them. It is a violation of transportation regulations to refill compressed gas cylinders without the express written permission of the owner.비 호환성
Water and air reactive Fluorides form explosive gases on contact with strong acids, acid fumes, and sodium. Water and air reactive. Corrosive. Fumes in air. Decomposed exothermically by water, alcohols, or moisture in the air to hydrofluoric acid and silicic acid, forming flammable and potentially explosive hydrogen gas. Attacks many metals in the presence of moisture.폐기물 처리
For laboratory quantities, transfer into an evaporating dish containing sodium bicarbonate, spray with ammonia (6M NH4OH)/6 M-ammonium hydroxide/while stirring and then spread with crushed ice. Continue spraying with ammonia until the smoke of ammonium chloride partly subsides and add iced water while stirring. Neutralize and slowly transfer the mixture into a drain with running water.실리콘 테트라플루오리드 준비 용품 및 원자재
원자재
준비 용품
실리콘 테트라플루오리드 공급 업체
글로벌( 60)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-66670886 |
info@dakenam.com | China | 15956 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| BEYOND INDUSTRIES (CHINA) LIMITED | +86-21-52699951; +8613917686115 |
sales@beyondindustriesgroup.com | China | 699 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | 0551-65418671 |
sales@tnjchem.com | China | 34572 | 58 |
| CD Chemical Group Limited | +8615986615575 |
info@codchem.com | China | 20356 | 58 |
| Shaanxi Didu New Materials Co. Ltd | +86-89586680 +86-13289823923 |
1026@dideu.com | China | 9020 | 58 |
| Henan Allgreen Chemical Co.,LTD | +86-37155567971 +86-13633837469 |
info@allgreenchem.com | China | 5986 | 58 |
| Mainchem Co., Ltd. | +86-0592-6210733 |
sale@mainchem.com | China | 32360 | 55 |
| PERIC Special Gases Co., Ltd. | 0310-7182720-613 15620672519 |
zhaoqingwei@pericsg.com | China | 40 | 58 |
실리콘 테트라플루오리드 관련 검색:
납 플루오로실리케이트 플루오린(불소) 규불화아연 칼슘 헥사플루오로실리케이트 규화불화 나트륨 플루오로규산 니켈 헥사플루오로실리케이트 실리콘 테트라플루오리드 동 실리코플루오라이드 헥사플루오르화규소산 리튬 바륨 헥사플루오로규산염 칼륨 헥사플루오로규산염 불화규산 카드뮴
Magnesium fluorosilicate
SILVER HEXAFLUOROSILICATE
Cryptohalite ((NH4)2(SiF6))
TRIFLUOROSILANE: 20-25% SOLUTION IN TETRAFLUOROSILANE,20-25% SOLUTION IN TETRAFLUOROSILANE
COBALT SILICOFLUORIDE