| Company Name: |
Wuhan kamik Technology Co., Ltd
|
| Tel: |
18062587608 |
| Email: |
201981259@qq.com |
| Products Intro: |
Cas:17341-24-1
ProductName:Lithium
Purity: 99% | Package: 5KG;1KG;25KG
|
| Company Name: |
Shanghai Sig Biotechnology Co., LTD
|
| Tel: |
021-57810052 18930344717 |
| Email: |
2089316240@qq.com |
| Products Intro: |
Cas:17341-24-1
ProductName:Lithium
Purity: 95% | Package: 25kg/
|
- Diquat ion
-

- $15.00 / 1KG
-
2021-08-12
- CAS:2764-72-9
- Min. Order: 1KG
- Purity: 99%+ HPLC
- Supply Ability: Monthly supply of 1 ton
- Diquat ion
-

- $15.00 / 1Kg/Bag
-
2020-11-16
- CAS:2764-72-9
- Min. Order: 1Kg/Bag
- Purity: 99%
- Supply Ability: 20 Tons
|
| | Lithium Ion Basic information |
| Product Name: | Lithium Ion | | Synonyms: | Lithium ion (1+);LITHIUM ION STANDARD;LITHIUM ION CHROMATOGRAPHY STANDARD;LITHIUM, ION CHROMATOGRAPHY STANDARD SOLUTION;Lithiumcation;Lithium, ion (Li1+);LITHIUM ION | | CAS: | 17341-24-1 | | MF: | Li+ | | MW: | 6.94 | | EINECS: | | | Product Categories: | | | Mol File: | 17341-24-1.mol | 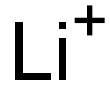 |
| | Lithium Ion Chemical Properties |
| Provider | Language |
|
ALFA
| English |
| | Lithium Ion Usage And Synthesis |
| Characteristics | Lithium Ion Batteries are characterized by higher specific energy, higher energy density, higher energy efficiency, a longer cycle life, and a longer calendar life.
| | Uses | Lithium Ion is a type of rechargeable battery that uses the reversible intercalation of Li+ ions into electronically conducting solids to store energy.
| | Hazard | Lithium Ion batteries can be a safety hazard if not properly engineered and manufactured because they have flammable electrolytes that, if damaged or incorrectly charged, can lead to explosions and fires.
| | Fire Hazard | Lithium-ion batteries can be a safety hazard since they contain a flammable electrolyte and may become pressurized if they become damaged. A battery cell charged too quickly could cause a short circuit, leading to overheating and explosions and fires.
| | Safety | The two main reasons for lithium-ion battery fires and explosions are related to processes on the negative electrode (cathode). During a normal battery charge lithium ions intercalate into graphite. However, if the charge is forced to go too fast (or at a too low temperature) lithium metal starts plating on the anode, and the resulting dendrites can penetrate the battery separator, internally short-circuit the cell, resulting in high electric current, heating and ignition. In other mechanism, an explosive reaction between the charge anode material (LiC6) and the solvent (liquid organic carbonate) occurs even at open circuit, provided that the anode temperature exceeds a certain threshold above 70°C.
|
| | Lithium Ion Preparation Products And Raw materials |
|