- 881677-11-8
-

- $300.00 / 100kg
-
2024-05-30
- CAS:881677-11-8
- Min. Order: 100kg
- Purity: 99.99%
- Supply Ability: 100Tons
|
| | 5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrole-3-carbaldehyde Basic information |
| Product Name: | 5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrole-3-carbaldehyde | | Synonyms: | 1H-Pyrrole-3-carboxaldehyde, 5-(2-fluorophenyl)-1-(3-pyridinylsulfonyl)-;5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrole-3-carbaldenhyde;5-(2-fluorophenyl)-1-(3-pyridinylsulfonyl)-1H-Pyrrole-3-carboxaldehyde;TAK-438( Vonoprazan fumarate) intermediate 4;(5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl)-N-methylmethanamine;Vonoprazan-026;TAK438 Impurity 67;Vonoprazan impurity 31/5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrole-3- carbaldehyde | | CAS: | 881677-11-8 | | MF: | C16H11FN2O3S | | MW: | 330.33 | | EINECS: | 807-719-6 | | Product Categories: | 881677-11-8 | | Mol File: | 881677-11-8.mol | 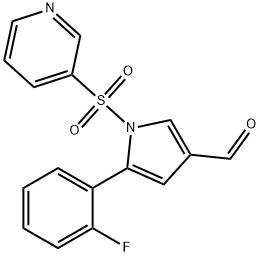 |
| | 5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrole-3-carbaldehyde Chemical Properties |
| Boiling point | 563.7±60.0 °C(Predicted) | | density | 1.37±0.1 g/cm3(Predicted) | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | solubility | Chloroform (Slightly), DMSO (Slightly) | | form | Solid | | pka | -0.34±0.11(Predicted) | | color | Pale Brown to Light Brown | | InChI | InChI=1S/C16H11FN2O3S/c17-15-6-2-1-5-14(15)16-8-12(11-20)10-19(16)23(21,22)13-4-3-7-18-9-13/h1-11H | | InChIKey | IXCSYEVJOAWXRH-UHFFFAOYSA-N | | SMILES | N1(S(C2=CC=CN=C2)(=O)=O)C(C2=CC=CC=C2F)=CC(C=O)=C1 |
| | 5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrole-3-carbaldehyde Usage And Synthesis |
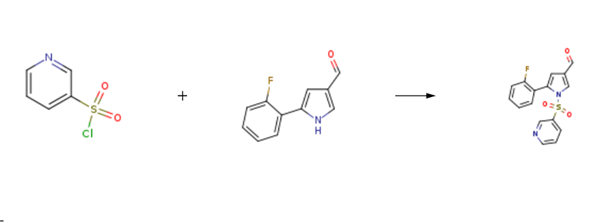
| Chemical Properties | Pale Brown to Light Brown Solid. | | Uses | 3-Des-N-methylmethanamine Vonoprazan-3-carbaldehyde is an impurity of Vonaprazan (V767010) a novel potassium-competitive acid blocker for the treatment of acid-related diseases. | | Synthesis | 5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrole-3-carbaldehyde is prepared by the reaction of 3-pyridinesulfonyl Chloride and 5-(2-fluorophenyl)-1H-pyrrole-3-carbaldehyde. The specific synthesis steps are as follows:
Take 10.0g (52.86mmol) of compound, 5.9g (58.15mmol) of triethylamine and 100mL of dichloromethane, cool the system to 0°C, add dropwise a dichloromethane solution (100mL) of compound G (58.15mmol), and add dropwise After completion, the system was reacted at room temperature for 6 hours, 100mL of water was added and washed twice, the organic phase was dried with anhydrous sodium sulfate, and concentrated under reduced pressure to obtain 16.4g of 5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrole-3-carbaldehyde, with a yield of 93.8%.
 | | Solubility in organics | Chloroform (Slightly), DMSO (Slightly) |
| | 5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrole-3-carbaldehyde Preparation Products And Raw materials |
|